
The programs will be administered by the FDA until 2028

The agency’s Center for Veterinary Medicine has set forth its goals for the 2024 to 2028 fiscal years

The agenda is a step toward the FDA modernizing its approach to evaluating and supporting creating innovative animal and veterinary products.

The agency is accepting public comments for 60 days from the date of publication in the Federal Register, starting September 15, 2023.

Fidoquel-CA1 is indicated to treat seizures associated with the condition

Multiple lots of Darwin’s Natural Pet Products raw cat and dog food have tested positive for Salmonella
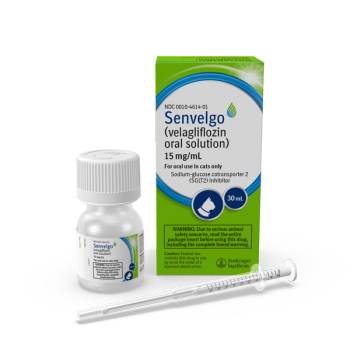
SENVELGO is a once daily medication to improve glycemic control in cats with diabetes mellitus

The administration is looking for suggestion on best framework for collecting this data

This approval is the latest addition of the NexGard portfolio

Midwest Veterinary Supply was also sentenced to 1 year probation for distributing “misbranded” pharmaceuticals

Apoquel Chewable tablets from Zoetis are indicated to help manage itching and inflammation caused by allergic dermatitis and atopic dermatitis

Status change of certain medically important antimicrobial drugs for animals from over-the-counter to prescription

The joint decision to expand the MRA's Sectoral Annex for Pharmaceutical Good Manufacturing Practices is a step toward strengthening use of each other’s animal drug inspection expertise and resources

Conditionally approved by the FDA, fuzapladib sodium for injection is a leukocyte function-associated antigen-1 activation inhibitor.

Zoetis’ bedinvetmab injection is a prescription solution targeting canine nerve growth factor.

The first drug for the control of nonregenerative anemia associated with chronic kidney disease in cats

The NexGard COMBO topical solution from Boehringer Ingelheim helps prevent tapeworms and other intestinal parasites as well as fleas, ticks, and heartworms.

The most recent first-time generic drug approvals by the FDA

Bimasone Injectable Solution is a corticosteroid drug that contains flumethasone in the same concentration and dosage form as Flucort.

This newly approved drug will help prevent acute vomiting and vomiting due to motion sickness

Tauramox is the first generic moxidectin injectable solution to control internal and external parasites
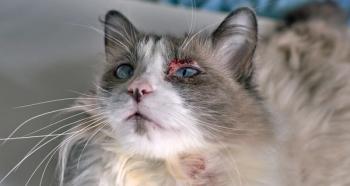
Modulis for Cats by Provetica contains the same active ingredient in the same concentration and dosage form as the Atopica for Cats brand product.

The pour-on NSAID for cattle is now FDA-approved for control of fever with mastitis

Tracey Forfa, JD, is the first nonveterinarian to hold the CVM directorship

The therapy is an antiparasitic drug that contains the same active ingredient as Dectomax.

We have compiled a countdown of our top 20 articles in 2022, here is number 1

We have compiled a countdown of our top 20 articles in 2022, here is number 4

We have compiled a countdown of our top 20 articles in 2022, here is number 7

We have compiled a countdown of our top 20 articles in 2022, here is number 8
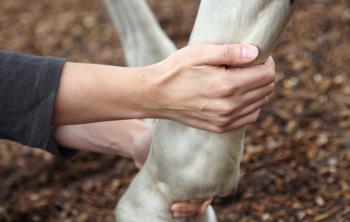
Appropriate management can help control the clinical signs and improve equine quality of life