A pain in the neck: diagnosing and treating neck and back pain (Proceedings)
Neck and back pain are common presenting complaints in veterinary medicine and occur in animals with a variety of signalments. Animals may present with a chronic history of lower-grade discomfort, although acute presentations of moderate to severe pain are common and extremely distressing to owner and pet alike.
Neck and back pain are common presenting complaints in veterinary medicine and occur in animals with a variety of signalments. Animals may present with a chronic history of lower-grade discomfort, although acute presentations of moderate to severe pain are common and extremely distressing to owner and pet alike. Pain may originate from a variety of locations associated with the spine, including vertebrae, nerve roots, meninges and possibly the intervertebral disk. A variety of etiologies are possible, although most patients fit within "the big 5" causes listed below.
Neck and back pain – the big five
1. Intervertebral disk disease (IVDD)
2. Diskospondylitis
3. Meningomyelitis
4. Trauma
5. Neoplasia
Neck and back pain – other etiologies
1. Lumbosacral disease (LSD)
2. Caudal cervical spondylomyelopathy (CCSM, "Wobbler's" syndrome)
3. Atlantoaxial subluxation (AAS)
4. Caudal occipital malformation syndrome (COMS, "Chiari-like" malformation)
5. Space-occupying intracranial disease (e.g., Brain tumor)
6. Spondylosis deformans (rare cause of neck/back pain)
7. Polyarthritis (rare cause of neck/back pain)
8. Polymyositis (rare cause of neck/back pain)
Diagnostic considerations for patients with spinal pain
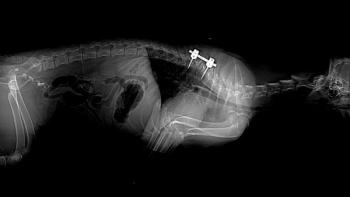
The diagnosis of the cause underlying neck and back pain revolves around imaging of the spine and spinal cord and examination of cerebrospinal fluid (CSF). Survey radiographs of the affected area are the typical starting point, and are usually diagnostic in cases of diskospondylitis, revealing lysis and malformation of the vertebral endplates next to the affected disk space. Discovery of diskospondylitis should initiate radiographic examination of the entire spine, as multiple lesions are often seen. Bacterial culture of the urine and blood and serum titers for infectious organisms (e.g., Brucella canis, Aspergillus spp.) may help to determine the offending organism and site of origin. Examination of urine sediment may reveal fungal hyphae in some Aspergillus infections. Fractures and luxations of the spine are usually seen with plain radiographs, although the changes may be subtle in some cases. Atlantoaxial subluxation is usually obvious, demonstrated by an increased distance between the dorsal arch of C1 and the dorsal spinous process of C2. Occasionally radiographs taken with the neck in mild flexion may facilitate the diagnosis, but should be obtained with extreme caution (particularly in anesthetized animals) to avoid further spinal cord damage. The dens (odontoid process) is often hypoplastic or absent in these animals, a finding which can also facilitate diagnosis. Neoplasms involving the bone may show lytic and/or proliferative changes on survey radiographs if a sufficient portion of the bone is involved. Animals with IVDD often show typical changes on radiographs, including calcified disks in situ, narrowed intervertebral disk spaces and radiodense material within the intervertebral foramen, although definitive diagnosis usually requires additional imaging techniques.
Additional diagnostic imaging techniques applicable to spinal disease include contrast studies (e.g., myelography, epidurography), nuclear scintigraphy, computed tomography (CT) and magnetic resonance imaging (MRI). Myelography demonstrates compressive spinal lesions, such as IVDD, CCSM, and neoplastic disease. A CSF sample should be collected before performing myelography, as the contrast material creates an inflammatory reaction, complicating later analysis. Myelography is rarely useful for LSD, as the spinal cord typically ends over L5-6 in the dog. Epidurography may show compression of the cauda equina, but is rarely used with the current availability of CT and MRI.
CT can be useful in LSD or after myelography to further delineate the location and character of compressive lesions. CT is much more sensitive than survey radiography, and may show obvious evidence of disease in animals with neoplastic lesions or diskospondylitis when radiographs are equivocal. MRI is superior to most other imaging techniques for soft tissue disease involving the spinal cord or nerve roots, and is indispensable for certain conditions, such as COMS.
Analysis of CSF is critical for making a diagnosis of meningomyelitis, and typically shows elevations in cell counts (pleocytosis) and protein levels. Specific serum titers or other tests (e.g., PCR) for infectious diseases may be considered for animals with meningomyelitis. Most compressive spinal cord disease will result in elevations of protein levels without pleocytosis. Occasionally neoplastic cells are seen in CSF, although this is rare. If AAS is a consideration, survey radiographs must be performed before collection of CSF from the cerebellomedullary cistern to avoid serious spinal cord damage.
Electromyography (EMG) and other electrodiagnostic tests are occasionally useful in the identification of focal spinal lesions or to demonstrate neuromuscular diseases (e.g., polymyositis).
Treatment considerations for neck and back pain patients
Compressive disease involving the spinal cord (e.g., IVDD, AAS, CCSM, LSD, fractures/luxations, neoplasia) and/or nerve roots is best treated with surgical decompression in many cases. Certain of these conditions, such as fractures, AAS and in some cases CCSM and LSD, require surgical stabilization as well. However, many patients with compressive diseases can be managed medically, usually with a combination of strict confinement, analgesics and occasionally glucocorticoids. NSAIDS are often ineffective for nerve root pain, although they may be useful for lesions of the spine itself. NSAIDS shouldnot be used in combination with glucocorticoids. Diazepam and methocarbamol are useful to control the pain associated with muscle spasm secondary to nerve root compression. Gabapentin can be very useful for pain of neuropathic origin, and tramadol also has a role in these patients. Ketamine and amantadine are NMDA receptor antagonists that can be useful adjuncts to address some of the pathophysiologic processes associated with pain ("windup"), and may be beneficial for acute and chronic pain patients respectively. Pregabalin is a relatively new medication with a mechanism of action similar to gabapentin, but with a greater potency. Experience with veterinary patients is relatively limited thus far, although preliminary results in some patients with neuropathic pain are encouraging. Acupuncture can also be a useful adjunct, particularly in chronic cases.
Appropriate antibiotic therapy (ideally directed by culture and sensitivity) for 6-8 weeks is required for diskospondylitis cases. Meningomyelitis may require specific antimicrobial therapy if an infectious etiology is identified. Patients without an apparent infectious etiology usually respond well to glucocorticoid therapy, which is tapered to the lowest controlling dose, and may eventually be discontinued in some cases. Additional immunosuppressive or cytotoxic drugs may be useful in animals not controlled with glucocorticoids alone, or intolerant of their side effects. Animals with neoplasia involving the spine, meninges, nerve roots or spinal cord may benefit from surgical debulking and radiation therapy, or chemotherapy in some cases.
Some medications beneficial in the therapy of neck and back pain in small animals
Diazepam 2-10 mg/animal PO q 8 hours
Methocarbamol 15-20 mg/kg PO q 8 hours
Gabapentin 3-10 mg/kg PO q 6-12 hours
Tramadol 1-4 mg/kg PO q 6-12 hours
Ketamine 0.5 mg/kg bolus IV, then 0.1-0.3 mg/kg/hour constant rate infusion
Amantadine 1.25-4 mg/kg PO q 12-24 hours
Newsletter
From exam room tips to practice management insights, get trusted veterinary news delivered straight to your inbox—subscribe to dvm360.