Newer options for medically managing refractory canine epilepsy
Seizures are the most common neurologic problem encountered in small-animal practice.
Seizures are the most common neurologic problem encountered in small-animal practice. Primary, or idiopathic, epilepsy—a disorder characterized by recurrent seizures for which an underlying structural cause is not found—is diagnosed in 25% to 40% of dogs with seizures.1,2 Affected dogs characteristically have an onset of seizures between 1 and 5 years of age, and successful management requires lifelong medical attention.
Antiepileptic drug therapy is the cornerstone of treatment for epilepsy, and phenobarbital and potassium bromide are the most commonly prescribed antiepileptic drugs in veterinary practice. Phenobarbital decreases seizure activity by potentiating the effect of the inhibitory neurotransmitter, gamma-aminobutyric acid (GABA), in neurons. Potassium bromide's antiepileptic mechanism of action is thought to involve hyperpolarization of the neuronal membrane by the movement of negatively charged bromide ion via GABA-activated chloride channels.
However, about 20% to 30% of epileptic dogs never attain satisfactory seizure control with these conventional antiepileptic drugs and are considered refractory to treatment.3 In addition, both drugs have a narrow therapeutic index and a propensity to cause marked side effects. Less than half of epileptic dogs receiving phenobarbital or potassium bromide maintain a seizure-free status without experiencing drug-related adverse effects.4 Side effects range from sedation, vomiting, polyuria, polydipsia, and polyphagia to more serious complications such as bone marrow suppression, hepatotoxicosis, and pancreatitis.
Historically, treatment options for dogs that have responded poorly to conventional antiepileptic drug therapy have been rather limited. Many of the common antiepileptic drugs used in people with epilepsy are not acceptable alternatives in veterinary medicine because of a lack of efficacy (valproic acid, oral diazepam) and the potential for toxicosis (lamotrigine).
However, over the last 20 years several novel therapies for epilepsy have been developed, and these new antiepileptic drugs have improved seizure control and resulted in fewer adverse effects in people. Pharmacokinetic studies on many of these new antiepileptic drugs have been performed in dogs, and the results support their potential application in canine epilepsy. In addition, several recently published reports describe the treatment of epileptic dogs with these medications. Thus, veterinary medicine is seeing an increase in the options available for managing refractory seizures in dogs.
In this article, I discuss the potential use of five newer antiepileptic drugs: felbamate, gabapentin, pregabalin, zonisamide, and levetiracetam. I also provide a tip on how you can help clients manage cluster seizures at home (see "Sidebar: Home care for cluster seizures").
GENERAL TREATMENT CONSIDERATIONS
Regardless of which drug is used, optimal treatment results are best achieved by adopting a systematic approach to seizure management (Figure 1). Although guidelines may differ among clinicians, I think that antiepileptic drug therapy is indicated for an animal that is experiencing more than one seizure a month, has cluster seizures irrespective of frequency, or has any history of status epilepticus. As a general rule, monotherapy is preferred because it tends to avoid complications that may arise from drug interactions and may also improve compliance by providing a simple treatment regimen.
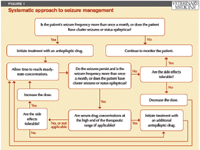
FIGURE 1: Systematic approach to seizure management
Adequate treatment response is assessed based on seizure frequency once steady-state serum drug concentrations are reached as well as on the severity of side effects. If seizures remain inadequately controlled (more than one seizure a month or the presence of cluster seizures or status epilepticus) in an animal with serum drug concentrations within the low therapeutic range and no evidence of medication-related side effects, the dosage of the antiepileptic drug can be increased. When drug concentrations reach the high end of the therapeutic range or unacceptable side effects develop and the animal still experiences monthly seizures or clusters of seizures, then the patient is considered refractory to that drug, and an additional antiepileptic drug should be included in the treatment regimen. This scheme can be followed when considering the use of any antiepileptic drug, including the conventional antiepileptic drugs as well as the newer drugs discussed below. Unless otherwise noted, all of the alternative seizure medications can be used in combination with both phenobarbital and potassium bromide.
ALTERNATIVE SEIZURE MEDICATIONS
Factors to consider when choosing an add-on medication include 1) mechanism of action, with preference given to drugs with a differing mechanism, 2) side effects, 3) the potential for drug interactions among antiepileptic drugs, 4) the required frequency of administration, which in turn may influence compliance, and 5) the cost. The important features of the new antiepileptic drugs approved for use in people being used extralabel in dogs are summarized in Table 1 and are discussed in more detail below.
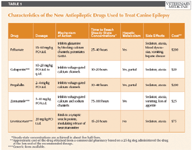
TABLE 1: Characteristics of the New Antiepileptic Drugs Used to Treat Canine Epilepsy
Felbamate
Felbamate was the first of the newer antiepileptic drugs to be approved in the United States for epilepsy in people. Side effects of aplastic anemia and hepatotoxicosis have since been reported in people, so its use has declined. The half-life of felbamate in dogs is five to eight hours, and the recommended oral dosage is 15 to 60 mg/kg every eight hours. Initiate treatment at the low end of the dosage range, and increase it as needed to control seizures. Felbamate serum concentrations are not routinely monitored, as is the case for many of the newer antiepileptic drugs. Rather, the drug is used to effect, and dosage adjustments are based on seizure frequency and side effects. Although most of the drug is excreted in the urine in dogs, some hepatic metabolism occurs, increasing the potential for drug interactions when phenobarbital and felbamate are administered concurrently.
One study has evaluated the use of felbamate as a sole antiepileptic drug in six dogs with partial onset seizures.5 Dosages ranged from 60 to 220 mg/kg/day, and all dogs showed reduced seizure frequency. Two dogs developed blood dyscrasias, characterized by thrombocytopenia, lymphopenia, and leukopenia, which resolved after the drug was discontinued. One dog developed keratoconjunctivitis sicca, although it was not determined that this problem was drug-related.
The use of felbamate as an add-on therapy has been reported in 16 dogs refractory to phenobarbital and potassium bromide.6 Twelve dogs had improved seizure control, but four of these dogs developed signs of liver dysfunction. Other side effects anecdotally reported in dogs receiving felbamate in combination with phenobarbital include sedation, nausea, and vomiting.7 It is recommended that complete blood counts and liver enzyme activities be measured every two or three months during treatment to assess for adverse effects. However, felbamate is used infrequently in veterinary patients because of the potential for side effects and drug interactions and because of the expense.
Gabapentin
Gabapentin was approved in the United States for use in people as an antiepileptic drug shortly after felbamate. Since its introduction, gabapentin has also been approved to treat neuropathic pain. In people, gabapentin is eliminated entirely by the kidneys, but in dogs it undergoes partial hepatic metabolism. The elimination half-life in dogs is two to four hours, requiring frequent drug administration to achieve steady-state concentrations. The recommended oral dosage is 10 to 20 mg/kg every six to eight hours.
Two studies have examined the use of gabapentin in refractory canine epilepsy. The first study involved 11 dogs administered gabapentin (10 mg/kg t.i.d.) in addition to phenobarbital and potassium bromide.8 A positive response to gabapentin, defined as ≥ 50% reduction in seizure frequency, was reported in six of the 11 dogs. The second study evaluated 17 dogs with refractory seizures that were administered gabapentin at a dose of 35 to 50 mg/kg/day divided twice or three times daily, also in conjunction with phenobarbital and potassium bromide (16 dogs) or phenobarbital alone (1 dog).9 This study found no significant decrease in the number of seizures over the study period for the entire population of dogs. However, seizures resolved in three dogs while they were receiving the medication. The side effects reported in both studies were sedation and ataxia.
The availability of generic gabapentin has made it affordable relative to other antiepileptic drugs used in veterinary patients. A liquid formulation (50 mg/ml concentration) of gabapentin exists, which facilitates the administration of lower doses to smaller patients. However, the liquid product contains 300 mg xylitol/ml, so it has the potential of causing adverse effects associated with the ingestion of this sugar alcohol.10 Consequently, I recommend that the tablet or capsule form of gabapentin be used preferentially in dogs.
Pregabalin
Pregabalin is a newer-generation drug in the same class as gabapentin. Limited information exists on its use in dogs as a treatment for epilepsy. Based on pharmacokinetic data in normal dogs, a dose of 2 to 4 mg/kg orally every eight hours has been recommended and was administered to six dogs with idiopathic epilepsy refractory to treatment with phenobarbital, potassium bromide, or both drugs.11 One dog was considered a drug failure, and four of the five remaining dogs had a mean seizure reduction of 59.3%. Five dogs exhibited side effects (sedation and ataxia) attributed to pregabalin.
Zonisamide
Zonisamide is a sulfonamide-derived antiepileptic drug introduced in the United States in 2000. Its half-life in dogs is 15 to 20 hours, which is relatively long when compared with the other new antiepileptic drugs, and it requires only twice-daily administration. Most of the drug is excreted unchanged by the kidneys, although some hepatic metabolism occurs. The dosage in dogs is 5 to 10 mg/kg given orally every 12 hours. The high end of the dose range is recommended when the drug is used in combination with phenobarbital since phenobarbital appears to facilitate zonisamide clearance.12 For this reason, it may also be helpful to measure serum zonisamide concentrations in dogs being treated concurrently with phenobarbital. A therapeutic range of 10 to 40 μg/ml has been suggested,13 which is similar to the therapeutic range in people. Zonisamide is a known teratogen in dogs, so its use should be avoided in pregnant animals.
Two recent reports on zonisamide as add-on therapy in dogs with refractory epilepsy demonstrated a favorable response in seven of 12 and nine of 11 dogs.13,14 Reported side effects included sedation, ataxia, and loss of appetite. A generic form of zonisamide is available, which has reduced the cost considerably since its introduction.
Levetiracetam
Levetiracetam (Keppra—UCB Pharma) is one of the more recently approved human antiepileptic drugs. Its unique mechanism of action is a potential advantage when the drug is used in combination with other antiepileptic drugs. Levetiracetam has minimal hepatic metabolism in dogs, with more than 80% of the drug excreted in the urine. The half-life in dogs is three to four hours, which necessitates frequent administration. The recommended oral dose is 20 mg/kg every eight hours.
A recent study evaluated levetiracetam as an add-on medication in dogs with idiopathic epilepsy that was refractory to phenobarbital and potassium bromide.15 Nine of 14 dogs responded, and the only side effect was sedation, which was observed in one dog. The main factor limiting the use of levetiracetam in dogs has been its expense, although a generic form of this drug has also recently become available.
Levetiracetam is also available in a parenteral formulation. One pharmacokinetic study has evaluated the disposition of the drug in six dogs after intravenous and intramuscular administration.16 A dose of 20 mg/kg results in desirable serum concentrations in a short period; with intramuscular administration, peak concentrations are reached in 40 minutes. Thus, this drug may prove useful in treating cluster seizures and status epilepticus in dogs, with the option of administering the drug intramuscularly if venous access cannot be obtained.
EFFICACY OF THE NEW ANTIEPILEPTIC DRUGS IN DOGS
When recommending a novel treatment, it is helpful if you can provide information on the expected effectiveness of the treatment, especially when the treatment involves a considerable investment of time or money on the pet owner's part. Unfortunately, extensive data of this nature are not available for the use of the new antiepileptic drugs in canine epilepsy. Rather, the information on drug use described above was obtained by administering the new antiepileptic drug in an open-label fashion: Dogs with poorly controlled seizures were given a new antiepileptic drug, and seizure frequency during administration of the new drug was compared with seizure frequency before the new drug was initiated. Because the studies were not placebo-controlled, the information obtained regarding drug efficacy may be inaccurate.
A reduction in seizure frequency during placebo administration has been documented in people with epilepsy,17 and, in my opinion, a similar phenomenon may occur in dogs. One likely cause for this placebo effect is regression to the mean, which is a statistical term used to describe the fluctuations that occur in biological variables over time and take the form of a sine wave around the mean. Epilepsy is a waxing and waning disorder, and fluctuations in seizure frequency are common over the course of the disease. Clients are most likely to seek a change in therapy for their pets when seizures are poorly controlled. Over time, improvement in the seizure frequency is probable, regardless of the treatment administered. However, this improvement may be erroneously attributed to a recently instituted change in therapy. Open-label studies cannot account for this bias, and, consequently, the efficacy reported in such studies may be falsely elevated.
Thus, from the information available to date, you can recommend a trial with a new antiepileptic drug, but, in my opinion, no representation should be made with respect to efficacy. As experience with these drugs grows in veterinary medicine and more research is performed on their applicability to dogs, additional information will become available to further guide recommendations on their use.
CONCLUSION
The management of refractory seizures can pose a considerable challenge to practitioners and pet owners. Ongoing patient follow-up is critical, and directed, appropriate therapy and monitoring should be used to optimize treatment results. With the approval of several new antiepileptic drugs for the management of human epilepsy over the last two decades, the treatment options available for our canine patients have also increased. Although our knowledge is still somewhat limited on these novel antiepileptic drugs, studies and clinical experience available to date suggest that they have the potential to improve seizure control and minimize adverse effects in affected dogs. Continued evaluation of these drugs in veterinary patients should provide additional details on how to use these therapies most effectively in managing canine epilepsy.
Editors' note: UCB Pharma provided levetiracetam for a clinical trial Dr. Muñana participated in.
Karen R. Muñana, DVM, MS, DACVIM (neurology)
Department of Clinical Sciences
College of Veterinary Medicine
North Carolina State University
Raleigh, NC 27606
REFERENCES
1. Podell M, Fenner WR, Powers JD. Seizure classification in dogs from a nonreferral-based population. J Am Vet Med Assoc 1995;206(11):1721-1728.
2. Berendt M, Gram L. Epilepsy and seizure classification in 63 dogs: a reappraisal of veterinary epilepsy terminology. J Vet Intern Med 1999;13(1):14-20.
3. Lane SB, Bunch SE. Medical management of recurrent seizures in dogs and cats. J Vet Intern Med 1990;4(1):26-39.
4. Podell M. Seizures in dogs. Vet Clin North Am Small Anim Pract 1996;26(4):779-809.
5. Ruehlmann D, Podell M, March P. Treatment of partial seizures and seizure-like activity with felbamate in six dogs. J Small Anim Pract 2001;42(8):403-408.
6. Boothe DM. Anticonvulsant therapy in small animals. Vet Clin North Am Small Anim Pract 1998;28(2):411-448.
7. Dayrell Hart B, Tiches D, Vite C, et al. Efficacy and safety of felbamate as an anticonvulsant in dogs with refractory seizures (abst). J Vet Intern Med 1996;10:174.
8. Platt SR, Adams V, Garosi LS, et al. Treatment with gabapentin of 11 dogs with refractory idiopathic epilepsy. Vet Rec 2006;159(26):881-884.
9. Govendir M, Perkins M, Malik R. Improving seizure control in dogs with refractory epilepsy using gabapentin as an adjunctive agent. Aust Vet J 2005;83(10):602-608.
10. Dunayer EK, Gwaltney-Brant SM. Acute hepatic failure and coagulopathy associated with xylitol ingestion in eight dogs. J Am Vet Med Assoc 2006;229(7):1113-1117.
11. Dewey CW, Cerda-Gonzalez S, Levine JM, et al. Pregabalin therapy for refractory idiopathic epilepsy in dogs (abst). J Vet Intern Med 2008;22:765.
12. Orito K, Saito M, Fukunaga K, et al. Pharmacokinetics of zonisamide and drug interaction with phenobarbital in dogs. J Vet Pharmacol Ther 2008;31(3):259-264.
13. Dewey CW, Guiliano R, Boothe DM, et al. Zonisamide therapy for refractory idiopathic epilepsy in dogs. J Am Anim Hosp Assoc 2004;40(4):285-291.
14. von Klopmann T, Rambeck B, Tipold A. Prospective study of zonisamide therapy for refractory idiopathic epilepsy in dogs. J Small Anim Pract 2007;48(3):134-138.
15. Volk HA, Matiasek LA, Reliu-Pascual AL, et al. The efficacy and tolerability of levetiracetam in pharmacoresistant epileptic dogs. Vet J 2008;176(3):310-319.
16. Patterson EE, Goel V, Cloyd JC, et al. Intramuscular, intravenous and oral levetiracetam in dogs: safety and pharmacokinetics. J Vet Pharmacol Ther 2008;31(3):253-258.
17. Burneo JG, Montori VM, Faught E. Magnitude of the placebo effect in randomized trials of antiepileptic agents. Epilepsy Behav 2002;3(6):532-534.
Newsletter
From exam room tips to practice management insights, get trusted veterinary news delivered straight to your inbox—subscribe to dvm360.