Clinical pathology of exotic companion mammal emergency presentations: The case of Daphne
Veterinary professionals can gain a lot of information on critical rabbit patients from history, physical examination, and diagnostic findings
Daphne, a 5-year-old female spayed rabbit, was brought to the emergency clinic with a recent history of decreased activity and fecal production and increased urination. The clinician covering emergency cases triaged Daphne as otherwise normal and sent her home with antibiotics for a suspected urinary tract infection and a critical care syringe feeding diet for possible gastrointestinal (GI) stasis.
Daphne presented again the following day and appeared dull and obtunded. The rabbit appeared to be dehydrated and had some mild abdominal splinting. The blood work results included as follows:
- CBC: moderate normocytic, normochromic anemia (28.4%; preferred range, 33%-50%); echinocytes, acanthocytes, and polychromasia
- Daphne also had lethargy, decreased fecal production, polyuria, dehydration, abdominal pain, elevated liver enzymes, anemia, and red blood cell morphology abnormalities.
GI disease in rabbits
Virtually any emergent condition in a rabbit can present with gastrointestinal signs. Disruption of the normal digestive process in rabbits can result in gastrointestinal dysmotility.
GI stasis is a vaguely defined term for decreased GI motility. Rabbit gastrointestinal syndrome (RGIS) has been proposed as a term in place of GI stasis to define the complex clinical signs, symptoms, and concurrent pathologic conditions affecting the digestive system of the rabbit.
Signs and symptoms of RGIS can include depression, decreased appetite increasing to anorexia, food and gas distention of the stomach and digestive tract (bloat), discomfort, the presence of uneaten cecotrophs (often referred to as diarrhea by owners), and true diarrhea. The onset of these signs can be sudden with rapid decline, so it is important to intervene quickly.
It should be kept in mind that RGIS is not a disease but a range of motility disorders with multiple potential primary and secondary causes. In many cases, rabbits respond to supportive care and the underlying cause remains undetermined.
Primary causes of RGIS
Dietary causes are a common primary etiology that incite RGIS. This includes abrupt change in diet, inappropriate food items (grains, large amounts of fruit), and diets generally low in fiber. Therefore, it is very important to perform an in-depth diet history when examining patients with RGIS symptoms.
Primary gastrointestinal disease caused by enteric pathogens is uncommon in rabbits but can include organisms such as Escherichia coli and rotavirus. Clostridial organisms are normal inhabitants of the rabbit GI tract and can proliferate under certain conditions, producing severe enteritis and in some cases enterotoxemia. Clostridial overgrowth is linked to low-fiber, high-carbohydrate diets, and administration of certain antibiotics (ie, oral penicillin, clindamycin, lincomycin, erythromycin).
GI parasites uncommonly produce disease, but coccidia can be a significant pathogen in young rabbits and those housed in overcrowded, stressful conditions. Other uncommonly reported diseases of the GI tract include toxin exposure (eg, plants, heavy metals) and coronavirus.
Foreign material implicated in obstruction includes bits of hair (trichobezoars), paper, fabric, and dried peas or beans. Other documented causes of obstruction include neoplasms, tapeworm cysts, abscesses, and adhesions. Cecal impaction is another unique disease presentation and is characterized by the presence of hard, firm material (usually food) in the cecum. It is frequently accompanied by stress and dehydration, but other underlying causes are uncertain.
Secondary causes of RGIS
To complicate matters, RGIS can occur secondary to any condition that can cause stress or illness in a rabbit, such as dental disease, trauma, any organ dysfunction, toxin ingestion, reproductive pathology, neoplasia, and infectious and inflammatory conditions.
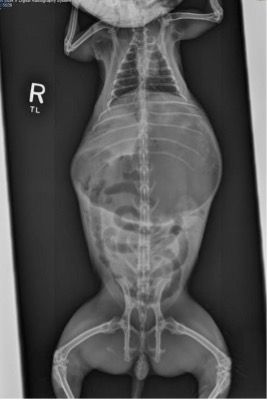
Figure 1: Rabbit with ileus
For example, anorexia secondary to any painful or debilitating condition reduces the amount of fiber for the digestive tract, resulting in GI hypomotility. Hypomotility can then result in alterations in cecal microflora with proliferation of pathogens, or overgrowth of naturally occurring Clostridium spp. If this chain reaction continues, worst-case situations can result in enterotoxemia and even death.
So when cases such as Daphne present, it is critical to express the importance of both supportive care and a full medical work-up to owners.
The rabbit in Figure 1 has generalized ileus (as opposed to obstruction) characterized by gas throughout the entire intestinal tract. At the time these radiographs were taken, the rabbit’s body temperature was 95 °F (35 °C) and it was hypotensive; note the decreased cardiac silhouette, typical with hypovolemia. Aggressive supportive care, including intravenous (IV) fluids with lidocaine constant rate infusion and thermal support, stabilized this rabbit and it was discharged uneventfully. A cause was never found.
Diagnosis of GI disease in rabbits
Physical examination findings depend on the extent of disease and the presence of other underlying disease processes. Rabbits can present from bright and alert with minimal significant findings to lethargic to moribund. Physical examination findings can include fluid imbalances (ie, dehydration, shock); abdominal distention, including gastric and intestinal distension; and gastric tympany. Other symptoms may be related to ongoing underlying conditions. Severely ill rabbits in hypovolemic shock are often hypothermic with pale mucous membranes and decreased capillary refill time with altered mentation (ie, depressed to comatose). Indirect blood pressure is low or unobtainable.
Basic diagnostic testing can be useful, especially in rabbits with other underlying disease processes such as renal disease. However, many rabbits with GI disease do not present with significant alterations in complete blood count and chemistry analysis. Anorectic rabbits may demonstrate elevations in hepatic enzymes due to hepatic lipidosis. More debilitated animals show alterations in blood urea nitrogen (BUN) and albumin consistent with dehydration. The most useful test for direct evaluation of the GI tract is high-quality radiographs of the abdomen in 2 views.
As primary bacterial gastroenteritis is uncommon in the rabbit, fecal culture and sensitivity are less useful in this species. The most useful application of this test may be rabbits with true diarrhea without a history of inappropriate diet or history of exposure to potential bacterial pathogens. Fecal floatation can be helpful in rabbits with diarrhea, especially in younger rabbits where primary coccidia is more common.
When ill rabbits present for examination, it is important to determine if RGIS is present, and if so, begin treatment and a diagnostic work-up to determine underlying contributing factors. Identification of an underlying cause is often difficult. In many cases, these patients respond positively to supportive therapy such as fluids, hand feeding, and motility enhancing drugs.
Some hospitalized rabbits may need collars placed to prevent them from chewing at IV catheters (Figure 2). Unfortunately, this can decrease food intake. Although multiple food options are available, it is important to provide supplemental nutrition via syringe feeding.
Figure 2: Collars for hospitalized rabbits

Blood glucose and sodium measurement in rabbits with RGIS
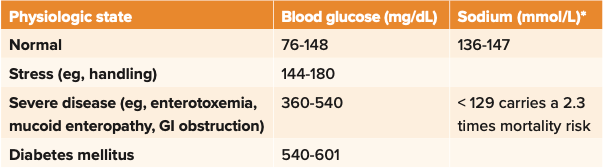
We can get important information from blood work in some rabbits with RGIS. For example, blood glucose can serve as a prognostic indicator.1,2 Blood glucose level in rabbits is related to food intake, stress level, and severity of clinical disease. Like cats, stressed rabbits have a higher glucose level than nonstressed rabbits. Completely anorectic rabbits have a higher blood glucose level than those with normal food intake or hyporexia. Severe hyperglycemia (> 360 mg/dL, 20 mmol/L) has been associated with conditions with a poor prognosis. Mean blood glucose value of 444.6 mg/ dL (24.78 mmol/L) has been found in rabbits with confirmed GI obstruction vs rabbits with nonobstructive RGIS of 153 mg/dL (8.5 mmol/L). Therefore, not only is blood glucose useful as an indicator or the severity of the animal’s condition, but it also helps differentiate RGIS vs GI obstruction.
Significantly higher mortality (2.3-fold increase) is associated with sodium levels below 129 mEq/L in rabbits.3 As pseudohyponatremia can be seen with a high blood glucose level or sodium level decrease in response to high glucose level (related to fluid shift and sodium dilution), it is important to simultaneously measure plasma sodium and glucose levels (Table 11).
Table 1: Rabbit blood glucose and sodium levels as prognostic indicators1
*Osmolarity and tonicity must be used to differentiate true hyponatremia from pseudohyponatremia.
What about lactate?
Although rabbits’ D-lactate levels are similar to those of other mammals, L-lactate levels are higher in healthy rabbits than in other mammals. It should be noted that lactate values were significantly different in rabbits when measured on portable analyzers vs blood gas analyzers: 6.9 (+/– 2.7) mmol/L on portable analyzers, 7.1 (+/– 1.6) mmol/L on blood gas analyzers.4 A single value on a rabbit with an emergent presentation has poor diagnostic or prognostic values, but serial L-lactate measurements can be valuable. Interestingly, L-lactate values were persistently low with minimal fluctuations in critically ill rabbits, whereas rabbits with a good prognosis had increased lactate values.5
Other important findings
A 2019 retrospective found that the odds of nonsurvival over a 15-day period for anorectic rabbits with BUN values greater than 24.74 mg/dL were 3 times that for rabbits with BUN values less than 14.0 mg/dL.6 Another investigation found that rabbits with hypothermia at admission had a risk of death before or within 1 week after hospital discharge 3 times that of rabbits without hypothermia. The odds of death doubled with each 1 °C (1.8 °F) decrease in admission rectal temperature.7 Keep in mind that these studies are reporting trends but this does not mean all severely azotemic and hypothermic rabbits will die. With aggressive supportive care measures, we can see rabbits with all signs pointing toward a poor prognosis make a full recovery.
So what about Daphne?
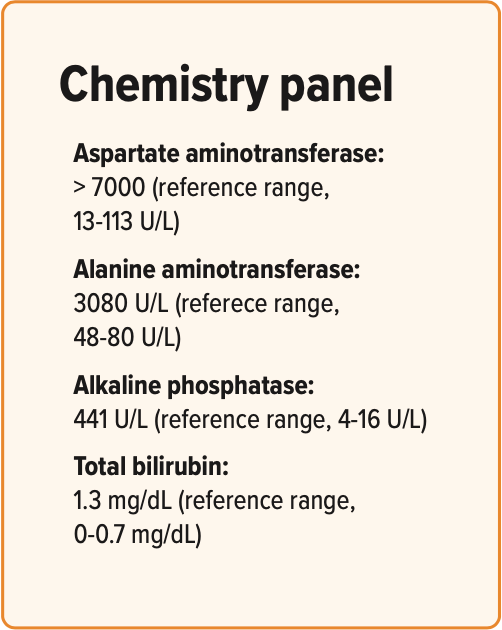
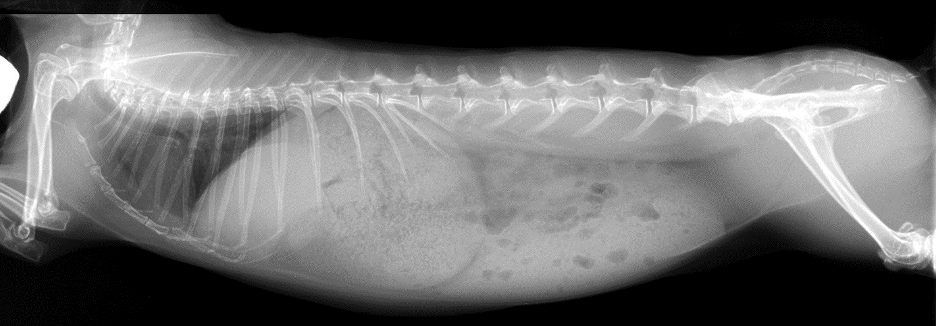
Are Daphne’s clinical signs and diagnostic findings indicative of a single problem or multiple? Further diagnostics were pursued to identify the underlying cause for Daphne’s RGIS, hepatopathy, and anemia. Radiographs were performed and revealed that the rabbit’s stomach was distended with apparently normal ingesta. Generalized ileus was noted, and Daphne’s hepatic shadow reported as normal or slightly enlarged (Figure 3). Since radiographs were not helpful in further understanding the hepatopathy, further imaging was pursued. Ultrasound revealed that her right medial liver lobe was hypoechoic with no apparent blood flow noted on color flow Doppler, and effusion was noted between the affected and nearby lobe. Daphne’s diagnosis was right medial liver lobe torsion.
Figure 3: Daphne’s radiograph
Liver torsion in rabbits
Liver torsion has been reported with increasing frequency in rabbits. The most common complaints on presentation include anorexia, lethargy, decreased fecal production, inappropriate urination and defecation, crouched or hunched body position, hiding behavior, and soft stools. These findings are typical for what is generally seen in RGIS, and most cases are initially suspected to be primary GI stasis. Because of this, it is recommended to perform diagnostics including blood work and radiographs.
Rabbits with liver lobe torsion will usually have some degree of anemia on complete blood count along with red blood cell fragmenting. Common serum biochemical abnormalities include elevated levels of alanine aminotransferase, alkaline phosphatase, and aspartate aminotransferase. Some rabbits can have other abnormalities such as elevated blood urea nitrogen and creatinine. Radiographs may show rounded liver margins or hepatomegaly, increased gastric or intestinal gas suggestive of gastrointestinal stasis or obstruction, loss of serosal detail, free peritoneal fluid, or an obscured liver border. Definitive diagnosis of liver lobe torsion in rabbits can be made with abdominal ultrasound. Common ultrasonographic findings include hepatomegaly or an abnormally large liver lobe, rounded lobar margins, mixed liver parenchymal echogenicity, hyperechoic perihepatic mesentery, and free peritoneal fluid. Color flow Doppler reveals a lack of or decreased blood flow in affected liver lobe(s).
Stabilization and prompt surgical removal of the torsed liver lobe is recommended to ensure the best outcome. Although not without risk, results of a study published in the Journal of the American Veterinary Medical Association showed that rabbits undergoing liver lobectomy had a longer median survival time than did rabbits that only received 8 medical treatment.
Some rabbits may survive with supportive care measures only, but survival rates may be less than 50%. Supportive care measures can include administration of subcutaneous or intravenous fluids, pain medications, supplemental feeding, prokinetic agents, and antibiotics. Surviving rabbits treated only with supportive care may be expected to have multiple episodes of recurrent gastrointestinal stasis within the first 1 to 2 months following diagnosis of hepatic torsion.8,9
Prognosis
Prognosis can be assessed based on a variety of factors. Rabbits with right liver lobe torsion were less likely to survive for 7 days than were those with caudate lobe torsion.8 Rabbits with moderate to severe anemia were less likely to survive for 7 days than were rabbits that were not anemic or had mild anemia. Other factors associated with a decreased 7-day survival rate were high heart rate at admission and additional days without defecation after admission. Use of tramadol was associated with an increased survival rate.8
So why is Daphne relevant to this discussion?
The plan for Daphne included stabilization in preparation for liver lobectomy the following day. Unfortunately, Daphne decompensated overnight and arrested. Cardiopulmonary resuscitation attempts were unsuccessful.
Although Daphne’s case had a sad ending, it illustrates the point that if
she had a diagnostic work-up on initial admission, there is a chance aggressive therapy instituted sooner could have changed her outcome and/or more quickly informed the seriousness of her condition. Daphne’s case has taught us that although it may be tempting to treat supportively or triage away for GI stasis without performing diagnostics, we may miss the cause of the patient’s illness.
Jennifer Graham, DVM, DABVP (Avian/Exotic Companion Mammal), DACZM, is a consultant at Moichor, an animal reference laboratory devoted to enhancing diagnostics using artificial intelligence and data. Graham graduated from Auburn University, completed an avian/exotic internship at the University of Georgia in Athens, followed by a residency in avian/exotic animal medicine at the University of California, Davis. Graham worked at Angell Animal Medical Center in Boston from 2006-2012 and ran the Zoological Companion Animal Medicine Service at the Cummings School of Veterinary Medicine at Tufts University in North Grafton, Massachusetts, from 2012 to 2022.
References
- Fisher P, Graham J. Rabbits. In: Carpenter J, Harms C (eds). Carpenter’s Exotic Animal Formulary, 6th ed. Elsevier; 2023:605-606.
- Harcourt-Brown FM, Harcourt-Brown SF. Clinical value of blood glucose measurement in pet rabbits. Vet Rec. 2012;170(26):674. doi:10.1136/vr.100321
- Bonvehi C, Ardiaca M, Barrera S, Cuesta M, Montesinos A. Prevalence and types of hyponatremia, its relationship with hyperglycemia and mortality in ill pet rabbits. Vet Rec. 2014;174(22):554. doi:10.1136/vr.102054
- Langlois I, Planché A, Boysen SR, Abeysekara S, Zello GA. Blood concentrations of D- and L-lactate in healthy rabbits. J Small Anim Pract. 2014;55(9):451-456. doi:10.1111/jsap.12247
- Ardiaca M, Dias S, Montesinos A, Bonvehi C, Barrera S, Cuesta M. Plasmatic L-lactate in pet rabbits: association with morbidity and mortality at 14 days. Vet Clin Pathol. 2016;45(1):116-123. doi:10.1111/vcp.12308
- Zoller G, Girolamo N, Huynh M. Evaluation of blood urea nitrogen concentration and anorexia as predictors of nonsurvival in client-owned rabbits evaluated at a veterinary referral center. J Am Vet Med Assoc. 2019;255(2):200- 204. doi:10.2460/javma.255.2.200
- Girolamo N, Toth G, Selleri P. Prognostic value of rectal temperature at hospital admission in client-owned rabbits. J Am Vet Med Assoc. 2016;248(3):288-297. doi:10.2460/ javma.248.3.288
- Ozawa S, Graham J, Guzman D, et al. Clinicopathological findings and prognostic factors of liver lobe torsions in domestic rabbits: 82 cases (2010-2020). J Am Vet Med Assoc. 2022;260(11):1334-1342. doi:10.2460/javma.22.03.0154
- Graham J, Orcutt C, Casale S, Ewing P, Basseches J. Liver lobe torsion in rabbits: 16 cases (2007-2012). J Ex Pet Med 2014; 23: 258-265.

Newsletter
From exam room tips to practice management insights, get trusted veterinary news delivered straight to your inbox—subscribe to dvm360.