Understanding tibial plateau leveling osteotomies in dogs
Cranial cruciate ligament pathology is a leading cause of lameness in dogs. Many surgical treatments have been described that aim to restore stifle joint stability and minimize the progression of subsequent osteoarthritis. Most surgical treatments seek to replace the function of the cranial cruciate ligament by substituting autologous tissues or synthetic materials. More recently, the tibial plateau leveling osteotomy (TPLO) procedure has been described, which alters the mechanical forces acting on the stifle, rendering the cranial cruciate ligament unnecessary.
Cranial cruciate ligament pathology is a leading cause of lameness in dogs. Many surgical treatments have been described that aim to restore stifle joint stability and minimize the progression of subsequent osteoarthritis. Most surgical treatments seek to replace the function of the cranial cruciate ligament by substituting autologous tissues or synthetic materials. More recently, the tibial plateau leveling osteotomy (TPLO) procedure has been described, which alters the mechanical forces acting on the stifle, rendering the cranial cruciate ligament unnecessary.1
The purpose of this article is to describe the clinical application of TPLO so that practitioners can better understand the surgical technique and explain the procedure and aftercare to clients. An exhaustive description of the complexities and nuances of the biomechanical basis, preoperative planning, and performance of a TPLO are beyond the scope of this article.
CHOOSING TPLO
Over time, TPLO has increased in popularity among veterinary surgeons. This increased popularity is based largely on subjective clinical experience as little hard data is available upon which to base such decisions and preferences.2-5 Some studies have suggested better limb function and less progression of osteoarthritis in patients treated with TPLO as compared with extracapsular stabilization.3,5 Other studies have failed to detect a difference between TPLO and extracapsular stabilization when a walking gait across a force plate was used as the outcome measure.4 Prospective, randomized, case-controlled studies using outcome measures meaningful to pet owners would be tremendously helpful but have not been conducted because they are challenging and expensive.2,5
Among those surgeons who regularly perform TPLO, patient selection varies widely. Some surgeons prefer the technique in all dogs regardless of age, size, or activity level. Others prefer the technique primarily in younger, large-breed, particularly active dogs. Veterinarians are encouraged to discuss these preferences with the surgeons to whom they refer their orthopedic patients.
BIOMECHANICS
The TPLO technique was developed from an understanding of active mechanisms that induce instability in a cranial cruciate ligament-deficient stifle. During weightbearing, ground reaction forces are resisted by the contraction of the extensor, or anti-gravity, muscles (quadriceps and gastrocnemius). These combined forces across the stifle compress the femur against the caudal- and distal-sloped proximal tibial plateau. The slope of the tibial plateau converts this femorotibial compression into a cranially directed shear force called cranial tibial thrust.6 This shear force does not normally induce cranial tibial translation in healthy canine stifles because it is constrained by the intact cranial cruciate ligament with probable contribution by the pull of the hamstring muscles on the proximal tibia. The magnitude of the cranial tibial thrust is a function of external ground reaction forces, internal muscular forces, and the slope of the tibial plateau. When cranial tibial thrust exceeds the tensile strength of a healthy cranial cruciate ligament or a weakened, degenerative cranial cruciate ligament, the ligament partially or completely ruptures.7
Several in vitro studies have evaluated the mechanical effects of the TPLO procedure.8,9 The tibial plateau angle in cranial cruciate ligament-deficient dogs within my practice varies from as little as 10 degrees to greater than 45 degrees, with most patients having a tibial plateau angle between 20 and 33 degrees. Leveling the tibial plateau to an angle of about 6.5 degrees reliably eliminates cranial tibial subluxation by converting cranial tibial thrust into caudal tibial thrust that must be constrained by the intact caudal cruciate ligament.8 Progressive leveling from 6.5 degrees toward 0 degrees further increases demand on the caudal cruciate ligament.8 Thus, overcorrection may increase the risk of subsequent caudal cruciate ligament failure, and undercorrection risks persistent stifle instability. A recent recommendation for the optimal postoperative tibial plateau angle is 5 degrees.7
Cranial cruciate ligament-deficient stifles also have increased internal rotation.10 In vitro studies have shown that TPLOs on cranial cruciate ligament-deficient stifles reduced internal rotation to near a physiologic level.8,10 Clinically observing some dogs after TPLO, however, reveals internal rotatory instability called a pivot shift.7,11 The pivot shift is an abrupt cranial shift of the lateral tibial condyle relative to the femoral condyle (internal stifle rotation). When you observe the gait of a dog with a pivot shift from behind, the stifle appears to suddenly buckle laterally. Torsional moments (twisting forces) acting on the stifle causing the pivot shift are likely the result of limb malalignment, some components of which can easily be corrected at the time of the TPLO if they are recognized and deemed significant by the surgeon. When a pivot shift is noted postoperatively in patients with good limb alignment, it often resolves clinically as the hamstring muscles develop in the postoperative rehabilitation period.
PREOPERATIVE RADIOGRAPHY
Precise patient positioning is required when obtaining radiographs in preparation for a TPLO.12 These radiographs are often obtained immediately before a TPLO because general anesthesia facilitates such precise patient positioning. The tibial plateau angle is measured on a mediolateral radiograph centered on the stifle joint but collimated to include the tarsus. If the stifle shifts away from the center of the radiographic beam, the tibial plateau angle will be measured incorrectly.12 For a true lateral radiograph of the stifle joint, the tibia must be parallel to the radiographic film (Figure 1).

Figure1. Patient positioning for a mediolateral radiograph of the stifle for TPLO planning. The dog is placed in lateral recumbency with the affected limb down. The stifle and hock are flexed to about 90 degrees. Note that the lateral surface of the hip, stifle, and hock are placed on the same level and are in contact with the radiographic cassette. The radiographic beam is centered on the stifle but includes the stifle and hock. The opposite limb is pulled cranially.
Radiographs obtained with the dog properly positioned display superimposition of the medial and lateral femoral condyles and a distinct medial tibial plateau, though this may be difficult to achieve in patients with marked femoral varus, valgus, or torsional deformity. The tibial plateau slope is defined by a line joining points at the cranial-most and caudal-most edges of the medial tibial condyle (Figure 2A). The tibial functional axis is defined by a line joining the midpoint between the intercondylar tibial tubercles (in the stifle joint) with the center of the talocrural joint. Next, a line is drawn perpendicular to the tibial functional axis. The tibial plateau angle is defined as the angle formed between the perpendicular line and the tibial plateau slope.
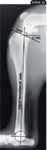
Figure 2A. A mediolateral radiograph showing the superimposition of the femoral condyles and the approximately 90 degrees of flexion in both the stifle and the tarsus. Note the lines demarcating the tibial plateau slope (TPS), tibial functional axis, and tibial plateau angle (TPA).
A recent geometric study of TPLOs emphasized the importance of surgical precision and suggested that extensive preoperative planning from radiographs may improve the surgeon's intraoperative performance.13 The surgeon can use transparent templates to evaluate the suitability of the various sizes of TPLO saw blades, jigs, and bone plates (Figures 2B & 2C). Because the position of the most proximal jig pin is critical, its desired position (immediately distal to the proximal point of the tibial functional axis and several millimeters distal to the subchondral bone plate) is marked on the film (Figure 2B). Then a template for the selected saw blade size, centered about the most proximal point of the tibial functional axis, is used to mark the desired position of the osteotomy on the film (Figure 2B).

Figure 2B. Templates corresponding to various sizes of TPLO saw blades can be superimposed on the mediolateral radiograph to select the proper size of blade and to plan its orientation centered about the most proximal point of the tibial functional axis (white arrow). The large dot at the intersection of the three lines (black arrow) indicates the proposed placement of the most proximal jig pin as close to the most proximal point of the tibial functional axis as the anatomy will permit.
Once the tibial plateau angle and appropriately sized saw blade have been determined, the amount of displacement needed along the circumference to accomplish the desired postoperative tibial plateau angle is determined from the Slocum TPLO chart (Slocum Stainless Steel TPLO Rotation Charts—Slocum Enterprises). A transparent template corresponding to different sizes of TPLO plates can be made from radiographic film. This template is used to plan the position of the bone plate and screws relative to the stifle joint and the proposed position of the jig and the osteotomy (Figure 2C)

Figure 2C. A transparent template corresponding to different sizes of TPLO plates is used to plan the position of the bone plate and screws relative to the stifle joint and the proposed positions of the jig and the osteotomy.
A caudocranial radiograph is used to evaluate the alignment of the tibia and to identify palpable landmarks that can help the surgeon avoid intra-articular placement of screws and jig pins. The stifle must be locked in full extension during caudocranial stifle radiography so that minimal physiologic rotation is present within the stifle joint (Figure 3A).
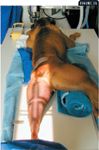
Figure 3A. Patient positioning for a caudocranial radiograph of the stifle for TPLO planning. The patient is positioned with the stifle fully extended. Abducting the opposite limb helps center the patella on the distal femur.
If the stifle is locked in full extension and is properly positioned (the patella is superimposed in the center of the distal femur and the fabellae are bisected by their respective femoral cortex), then the medial margin of the tuber calcis should be superimposed on the center of the distal tibia (Figure 3B). Medial or lateral deviation of the tuber calcis from this location indicates external or internal tibial torsion, respectively. The position of the fibular head with respect to the stifle articular surface on radiographs is correlated with palpation to help direct placement of the most proximal jig pin and subsequent proximal screws.

Figure 3B. A caudocranial radiograph showing the proper position of the stifle. Note the corresponding position of the medial margin of the tuber calcis on the center of the distal tibia, suggesting the absence of internal or external tibial torsion.
SURGICAL TECHNIQUE
Training and instrumentation
Many complications reported with this technique are related directly or indirectly to surgeon error.14-16 I developed experience with the technique in more than 700 procedures performed in private specialty practice and, then, mentored surgical residents as they gained expertise with TPLO in a university veterinary teaching hospital. The skills to expertly perform this technique consistently cannot be developed without solid orthopedic surgical skills, procedure-specific training, and an ongoing mentor relationship with a surgeon experienced in performing TPLOs. Aside from an extensive investment of time into training and mentoring, TPLO requires a considerable financial investment for surgical instrumentation. Necessary instruments include high-quality surgical lighting, suction and cautery equipment, a power drill, appropriate plating instruments and screw inventory, an osteotome and mallet, precision calipers, an array of retractors, and TPLO-specific components including a TPLO saw, saw blades, saw blade sharpeners, jigs, a drill guide, and implants. These combined training and instrumentation investments, in addition to increased operative time and complexity, account for the increased cost of this procedure compared with more traditional treatments.
Patient preparation
The affected hindquarter is shaved from the dorsal and ventral midlines to the mid-metatarsus, and the patient is positioned in dorsal recumbency. The foot is wrapped routinely; the wrap is kept distal to the hock so that the talocrural joint can be fully flexed and extended. The surgeon evaluates limb alignment and correlates palpation observations to the preoperative radiographs. Routine hanging limb preparation and surgical draping are then performed.
Surgical approach
A craniomedial approach to the stifle and proximal tibia is used. The surgeon makes a fascial incision starting between the sartorius muscle bellies proximally, extending distally through the tendinous insertion of the pes anserinus muscle group (caudal belly of the sartorius, gracilis, and semitendinosus muscles) about 6 to 10 mm caudal to the cranial margin of the tibial crest. At the distal end of the tibial crest, the fascial incision shifts to the cranial midline of the tibia to release the medial border of the cranial tibial muscle. Starting at the distal end of the surgical field, the surgeon lifts the semitendinosus muscle from the medial tibial surface to reveal the firm insertions of the gracilis and sartorius muscles on the medial aspect of the tibial crest. The shiny white insertion of the medial collateral ligament is visualized. The surgeon sharply incises the insertions of the gracilis and sartorius muscles, using care to protect the medial collateral ligament. Care is also used to preserve the pes anserinus muscle group layer as it is sharply incised from the underlying joint capsule.
Arthrotomy and meniscal procedures
Next, a medial parapatellar arthrotomy is performed routinely, and the cranial and caudal cruciate ligaments, medial and lateral menisci, long digital extensor tendon, synovial lining, and joint cartilage are evaluated. The cranial cruciate ligament is débrided as appropriate. Placing a small Hohmann retractor between the caudal cruciate ligament and the tibial plateau facilitates visualization of the menisci while protecting the caudal cruciate ligament from inadvertent laceration.
Medial meniscal pathology is often present and can vary from partial-thickness tearing to a full-thickness bucket-handle tear to a severe crushing injury. The surgeon should excise the pathologic segment of the medial meniscus, using caution to protect the caudal cruciate ligament in the process. Suction is essential, and headlight illumination is helpful. If the meniscus is intact, a medial meniscal release can be performed because it has been theorized to decrease the incidence of delayed meniscal injury (Figure 4). Alternatively, the medial meniscus can be released by transecting it at the midbody level as described later.
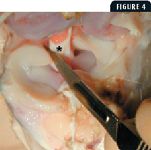
Figure 4. A stifle specimen. Note the position of the No. 11 scalpel blade for transecting the caudal tibial ligament of the medial meniscus to release the intact medial meniscus. The proximity of the caudal cruciate ligament (*) should be noted as it must be identified and preserved during the release procedure. Accidentally lacerating the caudal cruciate ligament negates the mechanical benefits of the TPLO.
TPLO preparation
In preparation for the TPLO, the surgeon takes great care to elevate the muscles from the caudal and lateral surfaces of the tibia to avoid lacerating the popliteal vessels. If indicated, the medial meniscus can be released at the midbody at this time (Figure 5). Next, radiopaque-marked gauze sponges are packed around the caudal and lateral tibial surfaces to retract the surrounding soft tissues (Figure 6).

Figure 5. A stifle specimen. Note the patellar ligament (*), the medial collateral ligament (#), and the caudal retraction of the pes anserinus muscle group (+). As an alternative to releasing the intact medial meniscus as shown in Figure 4, the medial meniscus can be released at the midbody by directing a No. 11 scalpel blade from the caudal margin of the medial collateral ligament toward the tubercle of Gerdy (at the cranial margin of the extensor groove of the tibia). This procedure is more difficult to visualize for the novice surgeon.
Jig placement
The jig (TPLO Jig—Slocum Enterprises) maintains alignment of the two bony segments after the osteotomy. Proper positioning and orientation of the most proximal jig pin are essential because the pin functions as the axis around which the tibial plateau rotates and as a visual guide for orienting the osteotomy. Improper positioning or orientation of the most proximal jig pin compromises subsequent aspects of the procedure.
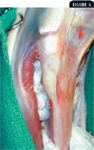
Figure 6. A stifle specimen. Gauze sponges are firmly packed along the caudal and lateral surfaces of the tibia to protect the surrounding tissues during the osteotomy.
The limb is precisely positioned, the jig is assembled, and a slow-speed, high-torque power drill is used to insert the proximal jig pin (Figure 7). After the pin is seated, it is cut flush with the surface of the jig to allow passage of the saw blade in later stages of the procedure.

Figure 7 To place the most proximal jig pin, the surgeon uses a slow-speed, high-torque power drill to insert the pin from medial to lateral so that the jig is parallel to the sagittal plane of the stifle. An assistant (in the background) guides the surgeon during pin placement so that the proximal jig pin is perpendicular to the sagittal plane of the stifle in both craniocaudal and proximodistal directions.
Next, the surgeon establishes a small soft tissue corridor for the distal jig pin. The surgeon inserts the distal pin in a similar manner as that used for the proximal jig pin, using care to keep it parallel to the proximal pin. Set screws are used to tighten the jig onto each of the jig pins (Figure 8). A small area of periosteum is elevated to prepare the bone for precise placement of the osteotomy.
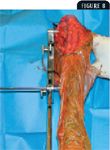
Figure 8. The surgeon's view of the TPLO jig while standing at the foot of the dorsally recumbent patient. Note the jig is parallel to the stifle's sagittal plane.
Osteotomy
An assistant places a small Hohmann retractor under the patellar ligament to retract it away from the saw blade. An appropriately sized TPLO biradial saw blade (Slocum Enterprises) is positioned in the desired location on the medial tibial cortex. The surgeon stabilizes the saw blade between the thumb and first finger until a reasonable kerf is established. Proper centering of the osteotomy kerf about the approximate proximal point of the tibial functional axis is confirmed. Surgical assistants help the surgeon keep the osteotomy blade centered about the proximal jig pin in all planes while cooling the saw blade with lactated Ringer's solution. The axis of the saw blade (like the most proximal jig pin) should be perpendicular to the stifle's sagittal plane in both the craniocaudal and proximodistal directions. Additionally, the saw blade should penetrate the caudal tibial cortex perpendicular to the tibial functional axis. Before the osteotomy is completed, the saw blade is removed, and a small, sharp osteotome is used to create reference marks to indicate the displacement needed along the osteotomy to accomplish satisfactory tibial plateau leveling (Figure 9). The osteotomy is then completed, and the gauze sponges are removed.
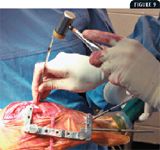
Figure 9. An osteotome and mallet are used to make reference marks on each side of the osteotomy to correspond with the desired amount of tibial plateau rotation.
The surgeon inserts an appropriately sized Steinmann pin temporarily into the plateau segment from cranial to caudal and slowly rotates the segment around the proximal jig pin until the reference marks line up (Figure 10). No effort is made to keep the medial surface of the tibial segments flush as this will typically create tibial malalignment.17 An appropriately sized Kirschner wire is passed across the osteotomy from the proximal-most end of the tibial tubercle into the tibial plateau to provide temporary stabilization during subsequent bone plate application. This Kirschner wire must penetrate the tibial tubercle proximal to the insertion of the patellar ligament to minimize the risk of tibial tubercle fracture.11 The surgeon performs the tibial compression test to ensure that cranial tibial thrust has been eliminated. The temporary Steinmann pin used for plateau rotation is removed.

Figure 10. A Jacobs chuck is applied to the temporary Steinmann pin so that the pin can be used as a handle, and the tibial plateau is slowly rotated around the proximal jig pin until the reference marks line up.
Next, the surgeon extends and flexes the stifle while evaluating the tibia for malalignment. Torsional malalignment is corrected by bending the distal jig pin either cranially or caudally as appropriate. Varus or valgus malalignment is corrected by sliding the jig in or out on the distal jig pin. A detailed discussion of limb alignment strategies is beyond this scope of this article. The edge of the saw blade is used to level out any stairsteps at the osteotomy site in the region of the proposed plate placement.
Bone plate application
The bone plate is contoured with bending irons for a precise fit against the medial tibial cortex to avoid inducing tibial malalignment. Cortical screws are placed through the fourth, fifth, and sixth plate holes into the distal tibial segment by using standard insertion methods. Next, cortical or cancellous screws (as appropriate) are placed in the second, third, and first holes, respectively. Provided no limb alignment alterations were made, these screws are inserted in compression mode to compress the osteotomy surfaces. If, however, tibial alignment corrections were made, the screws in the proximal segment are inserted in neutralization mode. Care is used to avoid placing these proximal screws into the stifle joint. The jig and temporary Kirschner wire are removed, and stifle stability, limb alignment, and implant placement are assessed (Figure 11).
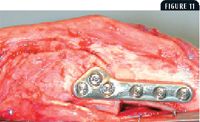
Figure 11. A surgical photo showing the TPLO plate holding the desired rotation of the tibial plateau after removal of the jig, temporary Steinmann pin, and Kirschner wire. Stifle stability, limb alignment, and implant placement are assessed.
The surgical field is lavaged thoroughly with lactated Ringer's solution. The joint capsule is apposed routinely with a continuous suture pattern. The popliteus muscle is apposed to the medial collateral ligament. The pes anserinus muscle group is apposed to the insertion remnant left on the medial margin of the tibial crest by using stifle flexion to ease tension during closure. Subcutaneous tissues are closed in one or two layers. Skin is apposed routinely. Mediolateral and caudocranial radiographs are obtained by using the same patient positioning described previously, and the tibial plateau angle is measured (Figures 12A & 12B). The radiographs are also evaluated for proper placement of the temporary jig pins (as evidenced by their pin tracts), osteotomy, and implants.

Figure 12A. A mediolateral postoperative TPLO radiograph that shows appropriate centering of the osteotomy in the approximate region of the most proximal point of the tibial functional axis. Note also that the proximal jig pin was placed on the tibial functional axis immediately distal to the stifle joint.
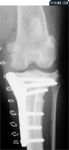
12B. A caudocranial postoperative TPLO radiograph that shows proper angling of screws such that intra-articular penetration is avoided.
PERIOPERATIVE CARE
Perioperative analgesia strategies should be both preemptive and multimodal. These strategies may include using systemic opioids such as morphine, nonsteroidal anti-inflammatory drugs (NSAIDs) such as carprofen, epidural opioids and local anesthetics such as morphine and bupivacaine, and N-methyl-D-aspartate receptor blockade agents such as ketamine at a continuous-rate infusion. Postoperative analgesia strategies may include oral administration of NSAIDs, opioids, or both and often will be tapered over two weeks.
Prophylactic use of cefazolin is recommended at a dose of 22 mg/kg administered intravenously 30 minutes before surgery and then every one-and-a-half to three hours during surgery thereafter. Assuming no breaks in sterile technique occur, cefazolin doses are repeated every eight hours during the first 24 hours after surgery. Little scientific support exists for the common practice of administering antimicrobials for several days postoperatively. Most patients that do not have complications are discharged from the hospital the day after surgery.
RECOVERY
Recovery from a TPLO consists of three stages: 1) incisional healing, 2) bone healing, and 3) physical rehabilitation. During the two-week incisional healing, it is imperative that the pet not be permitted to lick or chew at the incision because incisional dehiscence may increase the risk of osteomyelitis. An appropriate protective bandage, restraint collar, or both are recommended to prevent self-mutilation. It has been stated that fracture and osteotomy healing represents a race between bone healing and fixation failure. To this end, during the bone healing stage (usually eight to 12 weeks), patients should not be permitted to run, jump, or have free access to the yard, furniture, stairs, slippery floors, or other pets. Confinement to a small space with good footing and minimal external stimuli is advised when the patient is unsupervised (a large crate placed in a quiet room of the house works nicely). Short walks at a slow pace on a short leash several times a day are encouraged. Sling support of the hindquarters may be indicated in patients with multilimb disability, patients with poor strength or balance, and patients traversing slippery surfaces or steps. Typically, several weeks after surgery, the pet will feel rambunctious as limb function and comfort improve. This is an important time to remind pet owners that the bone is not yet healed.
Bone healing is monitored radiographically in four- to eight-week intervals until bony union is documented. Once bone healing is documented, then slow and methodical increases in activity can be instituted as part of the physical rehabilitation. This begins with increasing the length of leash walks each week, progressing to increased time off leash but under supervision. Anecdotally, too vigorous or too rapid a pursuit of reconditioning increases the risk of patellar desmitis. Physical rehabilitation improved the outcomes of patients treated with extracapsular stifle stabilization more than did surgery alone.18 Various protocols for physical rehabilitation are practiced for TPLO, but their benefits and risks have not been objectively identified. Empirically, carefully instituted physical rehabilitation by a trained therapist appears to hasten the recovery of limb use.
INTRAOPERATIVE AND POSTOPERATIVE COMPLICATIONS
A wide variety of complications have been reported for the TPLO.14-16 Intraoperative complications of extreme hemorrhage and intra-articular placement of screws or jig pins can be avoided with keen attention to detail. Hemorrhage from the popliteal vessels can be profuse. Familiarity with the cross-sectional anatomy of the proximal tibia and surrounding soft tissues helps prevent errant passage of the periosteal elevator, drill bits, taps, screws, and jig pins. Soft tissue complications in the postoperative period include seroma or hematoma, dehiscence, infection, and irritation from bandaging.
More severe complications, including tibial crest fracture, fixation failure, and patellar desmitis, are typically associated with a sudden and sustained increase in lameness and discomfort. Tibial crest fracture is fostered by the tension of the patellar ligament on the tibial tubercle that is unsupported caudally because of the rotation of the tibial plateau. This complication is rare when the osteotomy and the temporary Kirschner wire are properly placed and appropriate methods of tibial plateau rotation are used. However, the risk of tibial crest fracture appears to increase when the osteotomy is shifted too far cranially, when the tibial plateau is rotated excessively, when a gap is left between the rotated tibial plateau and the tibial crest, and when the temporary Kirschner wire is placed through the tibial crest distal to the insertion of the patellar tendon.
Failure of the plate fixation of the osteotomy may be the result of catastrophic loading of the limb in excess of the fixation strength or repetitive low-energy loading contributing to fatigue failure of the implants or implant loosening. Improper orientation of the osteotomy may increase the relative loading on the implants (less loading borne by the bone segments). Likewise, iatrogenic fracture of the fibula during a TPLO likely increases mechanical demand on the implants, thereby increasing the risk of failure. Osteomyelitis is often associated with increased lameness and the development of a draining tract or incisional drainage. Bacterial culture and antimicrobial sensitivity testing of fluids obtained by deep surgical site aspiration are used to select appropriate antimicrobial therapy. Once osteotomy healing is documented, implant removal (and repeat culture of the implant bed) may be indicated.
CONCLUSION
Cranial cruciate ligament pathology is a frequent cause of lameness in dogs, and surgical stabilization is the recommended treatment for most of these patients. While many surgical options exist, it is clear that TPLO is the preference of many surgeons for most patients. Early diagnosis, accurate radiographic measurement of tibial plateau angles, and surgical precision are key to achieving the desired functional outcome for each patient. It is hoped that the insights offered through this discussion will help your contributions toward your patients' recoveries.
Ross H. Palmer, DVM, MS, DACVS
Department of Clinical Sciences
College of Veterinary Medicine and Biomedical Sciences
Colorado State University
Fort Collins, CO 80523
REFERENCES
1. Slocum B, Devine T. Tibial plateau leveling osteotomy for repair of cranial cruciate ligament rupture in the canine. Vet Clin North Am Small Anim Pract 1993;23:777-795.
2. Aragon CL, Budsberg SC. Applications of evidence-based medicine: cranial cruciate ligament injury repair in the dog. Vet Surg 2005;34:93-98.
3. Schwarz PD. TPLO: a prospective clinical comparative study, in Proceedings. Am Coll Vet Surg Conf, 1999.
4. Conzemius MG, Evans RB, Besancon MF, et al. Effect of surgical technique on limb function after surgery for rupture of the cranial cruciate ligament in dogs. J Am Vet Med Assoc 2005;226:232-236.
5. Lazar TP, Berry CR, Dehann JJ, et al. Long-term radiographic comparison of tibial plateau leveling osteotomy versus extracapsular stabilization for cranial cruciate ligament rupture in the dog. Vet Surg 2005;34:133-141.
6. Slocum B, Devine T. Cranial tibial thrust: a primary force in the canine stifle. J Am Vet Med Assoc 1983;183:456-459.
7. Slocum B, Devine-Slocum T. Tibial plateau leveling osteotomy for cranial cruciate ligament rupture. In: Bojrab MJ, Ellison GW, Slocum B, eds. Current techniques in small animal surgery. 4th ed. Baltimore, Md: Williams & Wilkins, 1998;1209-1215.
8. Warzee CC, Dejardin LM, Arnoczky SP, et al. Effects of tibial plateau leveling on cranial and caudal tibial thrusts in canine cranial cruciate-deficient stifles: an in vitro experimental study. Vet Surg 2001;30:278-286.
9. Reif U, Hulse DA, Hauptman JG. Effect of tibial plateau leveling on stability of the canine cranial cruciate-deficient stifle joint: an in vitro study. Vet Surg 2002;31:147-154.
10. Arnoczky SP, Marshall JL. The cruciate ligaments of the canine stifle: an anatomical and functional analysis. Am J Vet Res 1977;38:1807-1814.
11. Palmer RH. Tibial plateau leveling osteotomy, in Proceedings. 10th Annu Am Coll Vet Surg Conf 2000;271-275.
12. Reif U, Dejardin LM, Probst CW, et al. Influence of limb positioning and measurement method on the magnitude of the tibial plateau angle. Vet Surg 2004;33:368-375.
13. Kowaleski MP, McCarthy RJ. Geometric analysis evaluating the effect of tibial plateau leveling osteotomy position on postoperative tibial plateau slope. Vet Comp Orthop Traumatol 2004;17:30-34.
14. Palmer RH. TPLO complications, in Proceedings. Am Coll Vet Surg Conf, 2004.
15. Priddy NH, Tomlinson JL, Dodam JR, et al. Complications with and owner assessment of the outcome of tibial plateau leveling osteotomy for treatment of cranial cruciate ligament rupture in dogs: 193 cases (1997-2001). J Am Vet Med Assoc 2003;222:1726-1732.
16. Pacchiana PD, Morris E, Gillings SL, et al. Surgical and postoperative complications associated with tibial plateau leveling osteotomy in dogs with cranial cruciate ligament rupture: 397 cases (1998-2001). J Am Vet Med Assoc 2003;222:184-193.
17. Wheeler JL, Cross AR, Gingrich W. In vitro effects of osteotomy angle and osteotomy reduction on tibial angulation and rotation during the tibial plateau-leveling osteotomy procedure. Vet Surg 2003;32:371-377.
18. Marsolais GS, Dvorak G, Conzemius MG. Effects of postoperative rehabilitation on limb function after cranial cruciate ligament repair in dogs. J Am Vet Med Assoc 2002;220:1325-1330.
Newsletter
From exam room tips to practice management insights, get trusted veterinary news delivered straight to your inbox—subscribe to dvm360.