Surgical drains are useful in small animal wound management
Practitioners must consider drain types, mechanism, monitoring, removal, and more
Content submitted by Thrive Pet Healthcare, a dvm360® Strategic Alliance Partner
The use of surgical drains in small animals has been an extremely useful, dependable method that allows uncomplicated wound healing. There are 2 basic types of drains most used in small animals: passive and active. Additionally, negative pressure wound therapy (NPWT) has become a reliable and more readily available tool for small animal practitioners. The use of drains and NPWT aid the practitioner in removing fluid and air between tissues.
The benefits of evacuation of serum, blood, and inflammatory debris and the reduction of swelling and pain can be better managed in many cases. A strict adherence to Halsted principles is advised when considering drain use and any form of wound closure. In some cases of contaminated wounds, daily management and serial debridement likely are necessary to achieve an environment suitable for final wound closure and drain placement.
Drain types
Drain materials are of varying types and may predicate use in certain situations. Common drain materials include latex rubber, polyethylene, polyvinyl chloride, silicone rubber, and red rubber.
Of these, the inflammatory response created by latex and red rubber must be considered when rationalizing use prior to implementation. Latex is softer, may be more comfortable, and may cause less tissue damage. Polyvinyl chloride drains should not be sterilized by use of ethylene oxide due to the binding of ethylene oxide and resultant hemolysis that may occur. Silicone is highly biocompatible and can be steam sterilized.1,2
All drains, regardless of composition, are recognized by the body as foreign material and will initiate an inflammatory response. Furthermore, drains require an exit from tissues and skin. Therefore, natural host defenses are compromised, albeit minimally, during drain use. This must be appreciated in the maintenance and care of drains to prevent migration of nosocomial organisms into the tissue being drained.
Drain mechanism
Passive vs active
This contrasts with active drains, which create pressure gradients allowing exudate to flow into and through the lumen of the drain. Collection of exudate in passive drains is typically done via an aseptically applied bandage, whereas collection when using active drains occurs in a reservoir. This reservoir is likely the portion of active drains that is used to create the pressure gradients promoting exudative flow.
Passive drains
In veterinary medicine, the most-used open, passive drain is the Penrose drain. Penrose drains are made of soft, pliable latex that is radiopaque. These drains are inexpensive and available in a variety of sizes. They are tubular in design, but drainage occurs on the outer surface area of the drain.1 The mechanism for drainage involves factors such as capillary action, gravity, overflow, and body movement or pressure between tissue layers. Consequently, Penrose drains should not be fenestrated nor should they be used in conjunction with suction. They are also not suitable for use in the thoracic or abdominal cavities because of the potential for retrograde flow of air and other fluids into the cavities.
Figure 1: Example of a Penrose drain exit in a gravity-dependent area separate from the suture line.
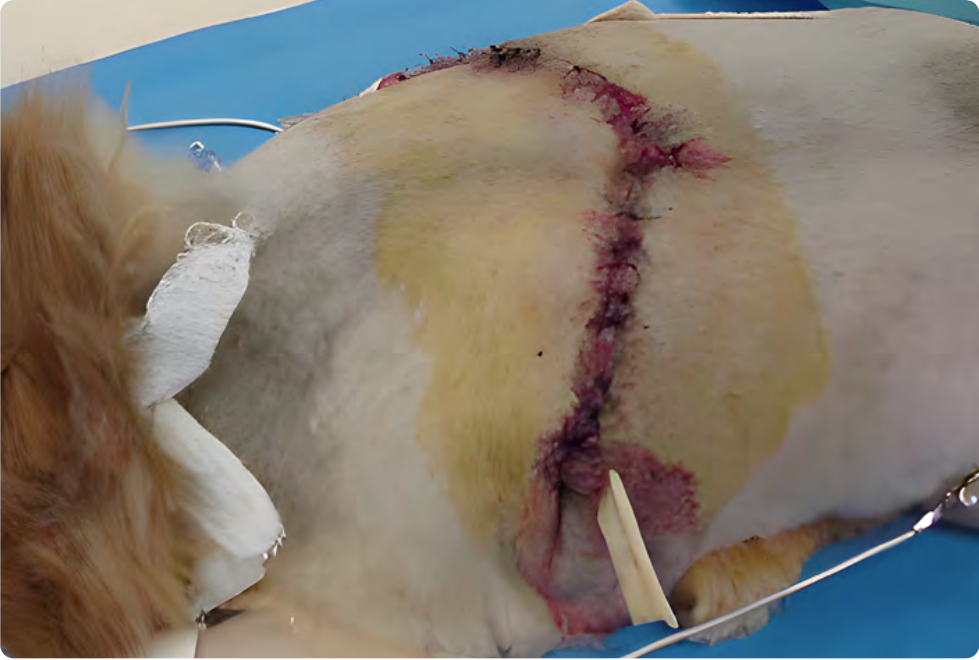
Typically, a Penrose drain is placed by positioning its deepest portion in the deepest aspects of the tissue defect or wound bed and exiting the distal aspect of the wound drain in a ventral gravity-dependent location (Figure 1). The exit hole of the Penrose drain should be large enough so the drain does not create an obstruction of exudate flow out of the wound.
The drain is kept in place through proper tissue layer closure over the latex, and 1 or 2 simple, interrupted, nonabsorbable sutures are placed through the drain and into the skin at the exit hole.1 Tacking the proximal aspect of the Penrose drain can only be considered with caution, as this can create a natural tension causing potential for drain breakage deep inside the wound.
Placing a Penrose drain and a loop with double exit holes on either end of the drain is unlikely to increase drainage significantly. This also creates an additional potential site for the entry of bacteria while the drain is in place. Absorbent sterile dressings and aseptic technique should be utilized over the external portion of the drain so that exudate can be evaluated to potentially reduce migration of bacteria into the wound bed. In addition, excoriation of skin around the drain exit is minimized. These dressings and the gauze used at the exit site can be secured by a variety of bandage techniques depending on the anatomical location of the drain.3
Closed active drains
Figure 2: Examples of the vacutainer with butterfly catheter and syringe with hypodermic needle in the hub and butterfly catheter to create a petite wound drain.

Closed active (or active suction) drains consist of a tube with a suction device attached to the exterior end of the drain. This suction device creates
a vacuum or pressure gradient that allows fluid to be pulled into the fenestrations typically located on the end of the drain located within the wound bed. The exudate then travels into the tube lumen and is finally collected into the suction device or reservoir. The reservoir is used for collecting fluid, maintaining suction, and allowing for quantification of exudate and characterization of fluid.
Closed active drains are preferable to passive drains for preventing bacterial migration into the wound bed and surrounding tissues. Due to the pressure gradients created by closed active suction drains, tissue adherence is improved compared with passive drains.1 Some examples of drain reservoirs may include the compressible “grenades” of Jackson-Pratt drain systems: vacutainers for homemade petite wound drains, syringes with needles passed through the plunger as desired suction was achieved, and rigid spring-loaded collection systems (Figure 2).
The fenestrated components of these closed active drain systems are available commercially or can be created using other readily available items (eg, butterfly catheters). If creating fenestrations in a drainage tube, the fenestrations should measure less than one-third of the tube diameter to prevent breaking or kinking of the drain tube. Additionally, to ensure appropriate function of the active system, all fenestrations should be in an airtight portion of the wound or tissue layers being drained. The exit of the drain should also be airtight to avoid air leakage or entry into the wound. Occasionally, bandages may help support this airtight seal along the closed wound margin, incision, or drain exit.2
Figure 3: Example of exit creation and tunneling by use of forceps for active drain tubing.
Figure 4: Example of non–gravity-dependent exit near the dorsum of an active drain.
When placing the drain tubing during wound or incision closure, the exit of a closed active system should be near the incision or margin of the wound closure (Figure 3). It should exit along the lines or within the plane of wound closure, as this may provide an entry point for bacteria or may result in dehiscence and additional drainage along the suture line.2 Because pressure gradients created along the length of the drain exit points are not gravity dependent (Figure 4), these exits can be made in locations that may facilitate drain maintenance and bandaging.1
To place these drains, often Halsted or Kelly forceps are tunneled from the surgical wound subcutaneously to the planned exit point. A small stab incision is then created over the tip of the forceps such that only the tip of the forceps and the diameter of the drain can be passed through the incision. Instead of passing the drain in retrograde fashion, a second similarly sized forceps can be passed in retrograde fashion, and the distal end of the drain is grasped and pulled from the wound to the outside to prevent contamination by natural flora and the skin surface. Commercially available drains may provide a trocar on the distal end, which facilitates tunneling through tissues for drain tube placement.
As with passive drains, securing the most proximal aspect of the drain tube in the wound or to the skin surface is not recommended. This may compromise the function of an active drain by allowing air into the wound as well as creating an opportunity for drain breakage. I prefer to secure the drain by using a purse string suture at the location of the drain exit along with a finger trap suture using nonabsorbable monofilament suture material.
Following drain tube placement, closure of the wound or incision is performed before attaching the reservoir so that active suction can be created. Dressings can then be applied to secure excessive tubing and the reservoir. Bandages may also protect against bacterial exposure and migration into the wound. Ideally, the veterinarian or staff performs management of the drain to avoid contaminating the drain tube and reservoir.1,4
Monitoring and removal
Many factors may determine how frequently dressings or bandages need changing. Initially, passive drain dressings and bandages may need to be changed once to twice daily to prevent skin maceration around the drain exit from the exudate. Similarly, closed active systems may require reservoir emptying every 1 to 2 hours prior to achieving decreases in the exudate collected.
Figure 5: “Grenade”-style collection reservoir for active drains.
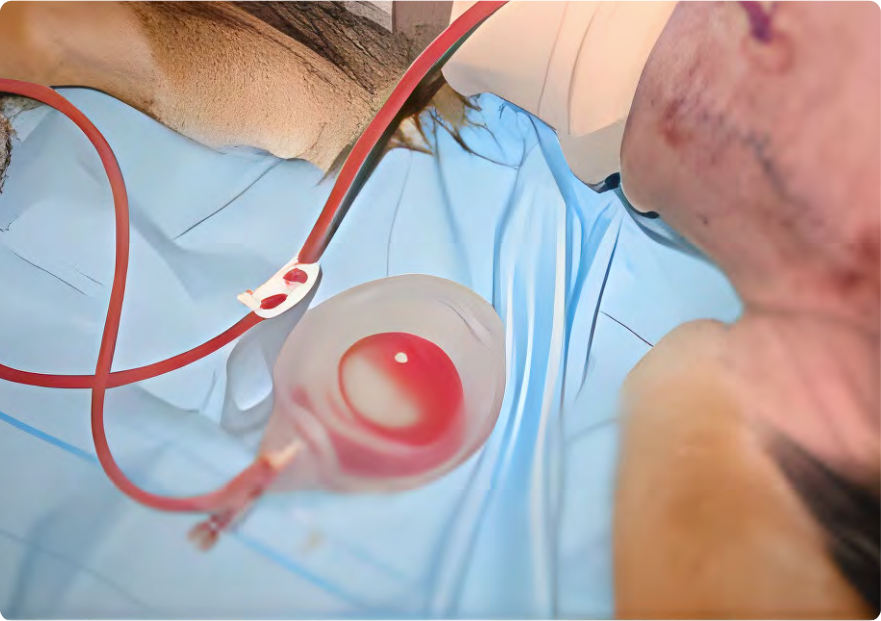
As tissues heal, exudate should become more serosanguineous and decrease in volume. Closed active systems will allow for the collection of fluid and for serial cytology to be performed to further evaluate changes in wound exudate.1 This may allow for the refinement of antimicrobial selection (Figure 5) if needed. Wound exudate eventually plateaus but is unlikely to cease entirely because the materials for any drain, active or passive, will still likely incite a fundamental inflammatory response due to the foreign material within the body.
When removing drain sutures, holding drains at the site of the exit wounds are typically cut, and the drains are removed aseptically and slowly. The most proximal end of the drains can be cut and provided for culture, should this be considered necessary. The drain exit sites are considered contaminated wounds and therefore are allowed to heal by secondary intention.4
NPWT
Figure 6: Occlusive bandage with foam and tubing used for negative pressure wound therapy.
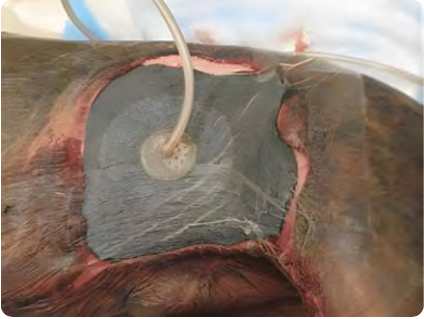
NPWT is a more elaborate form of a closed active drain system that uses precisely controlled intermittent suction to provide subatmospheric pressures at the wound surface.5 NPWT provides wound drainage but also may enhance the development of early granulation tissue by stimulating fibroplasia and fibroblast migration along with enhanced angiogenesis in the wound bed.5
A commercially available course, open cell foam is applied directly to the wound surface. This is coupled with a specialized collection disc and wound suction tubing overlaid by an occlusive wound dressing to form an airtight seal (Figure 6).
NPWT suction is created by using commercially available suction pumps with various settings. The reservoir is attached to the tubing and to the NPWT suction pump. Pump settings are considered most ideal from –65 mm Hg (for use in wound grafting) to –125 mm Hg for general wound suction.
Because of the foam’s unique nature and occlusive dressings, a variety of anatomical locations are suitable for NPWT (Figure 7). It is contraindicated for use in wounds with copious amounts of necrotic or devitalized tissue, coagulopathies, in thoracic wounds, or directly over neoplastic tissue, joints, and major vessels.5
Figure 7: Occlusive bandage for negative pressure wound therapy in a freshly debrided wound bed.

Additional considerations
Drains may be beneficial but are fundamentally foreign materials placed in wounds and healing tissue. When using drains, care must be taken to avoid opportunities for wound contamination. To reduce risk, use the fewest number of drains and exits (ideally 1 exit per drain) possible, adhering to Halsted principles when debriding and closing wounds. Appropriately manage drains following placement and remove them as soon as drainage is minimized. Care should also be taken to avoid placement of drains directly onto or immediately near healing luminal structures, neurovascular structures, or any tissue that may be damaged throughout drain use or removal.
Lastly, drain placement in association with the resection of neoplasms or undiagnosed masses would be considered a potential risk for tumor seeding along the drain path. This, however, remains to be proven.1 In my opinion, if the benefits of drain placement outweigh the risks of tissue closure without a drain, then drain placement may be a viable option.
Conclusion
Drain use along with considering the benefits and contraindications of drain type are helpful in small animal surgery. Veterinary practitioners can leverage
the benefits of drain use to provide successful outcomes with challenging wound closures when fluid accumulation and tissue adherence could be detrimental. By minimizing complications and promoting the aseptic management of drains along with careful monitoring, drains can provide additional utility in wound management.
Jude T. Bordelon, DVM, MS, MBA, DACVS-SA, is the medical director at Baton Rouge Veterinary Referral Center in Louisiana and Thrive Pet Healthcare’s national director of surgery. A native of Louisiana, he earned his undergraduate degree and doctor of veterinary medicine degree from Louisiana State University. He went on to complete a 1-year rotating internship in small animal medicine and surgery and a 3-year residency program in small animal surgery while pursuing a master of science degree at Oklahoma State University. After completing his residency, he remained at Oklahoma State University for 2 years as an associate professor in small animal surgery. He has since worked in private practice and academia.
References
- Campbell BG. Bandages and drains. In: Tobias KM, Johnston SA, eds. Veterinary Surgery: Small Animal. 2nd ed. Elsevier; 2018:221-230.
- Miller CW. Bandages and drains. In: Slatter DH,ed. Textbook of Small Animal Surgery. 3rd ed. Elsevier; 2003:244-249.
- Pavletic MM. Wound drainage systems. In: Pavletic MM. Atlas of Small Animal Wound Management and Reconstructive Surgery. 3rd ed. Wiley-Blackwell; 2010:52-60.
- Bristow PC, Halfacree ZJ, Baines SJ. A retrospective study of the use of active suction wound drains in dogs and cats. J Small Anim Pract. 2015;56(5):325-330. doi:10.1111/jsap.12339
- Stanley BJ. Negative pressure wound therapy. Vet Clin North Am Small Anim Pract. 2017;47(6):1203-1220. doi:10.1016/j.cvsm.2017.06.006

Newsletter
From exam room tips to practice management insights, get trusted veterinary news delivered straight to your inbox—subscribe to dvm360.