Thymoma in an 11-year-old dog: Internal medicine perspective
Dr. Sarah Schmid provides the internal medicine perspective on this thymoma case.
Dr. Sarah SchmidThymoma has been associated with many paraneoplastic syndromes, including myasthenia gravis,1 hypercalcemia,1,2 exfoliative dermatitis,3 erythema multiforme,4 T cell lymphocytosis,5,6 myocarditis,1,7 immune-mediated diseases (anemia, thrombocytopenia),1,8 and concurrent neoplasia.4,8,9 These paraneoplastic syndromes may occur at presentation, later in the disease course or even after tumor removal.10 The most common and well-established paraneoplastic syndrome is myasthenia gravis.
Myasthenia gravis has been reported in up to 47% of dogs with thymomas.8,11 However, when looking at dogs with myasthenia gravis, only 4.8% have a thymoma.12 In cats the opposite is true, with 4.7% to 6% of cats with a mediastinal mass having myasthenia gravis.13 However, if a cat has myasthenia gravis, a thorough workup should be performed to screen for a mediastinal mass, because it has been reported that as many as 52% of cats with myasthenia gravis have a mediastinal mass.14
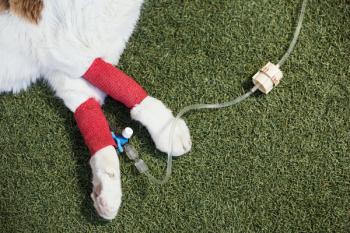
Figure 1. The patient displayed hypersalivation after developing megaesophagus.Myasthenia gravis is a neuromuscular disease characterized by muscle weakness. Some dogs may have a more focal form, in which only a small group of muscles are affected, resulting in megaesophagus (Figure 1), pharyngeal paralysis or a decreased palpebral reflex. Other dogs have generalized weakness that worsens with activity.15 Myasthenia gravis is considered an autoimmune disease because it results from the formation of autoantibodies against nicotinic acetylcholine receptors on the postsynaptic muscle terminal. These autoantibodies lead to complement-mediated destruction of the acetylcholine receptors, resulting in a diminished ability for muscle to respond to acetylcholine released into the synaptic cleft.
The gold standard for diagnosis of myasthenia gravis is the demonstration of serum autoantibodies against muscle acetylcholine receptors.16 Immunoprecipitation radioimmunoassay has high sensitivity (98%) and specificity (100%) for diagnosing myasthenia gravis.15
The treatment of choice for thymomas is surgical resection, and it is recommended whenever possible. In masses that cannot be surgically resected, radiation therapy can be considered. Although it may resolve with the removal of the thymoma, concurrent treatment of myasthenia gravis is often necessary. Treatment of myasthenia gravis focuses around increasing the amount of acetylcholine available in the neuromuscular junction by administration of long-acting anti-acetylcholinesterase drugs, such as pyridostigmine or neostigmine.15 In addition, treatment should include treating complications associated with myasthenia gravis, such as megaesophagus and aspiration pneumonia.
Treating megaesophagus involves small, frequent meals fed in an upright position.17 Because metoclopramide and cisapride are both smooth muscle prokinetic agents, they are thought to have no effect on the striated muscle of the canine esophageal body and, thus, are of unproven benefit in the treatment of megaesophagus.18 Should aspiration pneumonia result secondary to megaesophagus, an airway wash for a sample for culture and sensitivity should be performed if possible. A recent consensus statement provides antimicrobial guidelines for the treatment of respiratory tract disease in dogs and cats.19
The prognosis for dogs with surgically resectable (benign) thymomas without megaesophagus is good. In a study, dogs without megaesophagus and a resectable tumor had a one-year-survival rate of 83%.1 If megaesophagus is concurrently present, the patient's prognosis is thought to be guarded to poor, as many of these dogs succumb to recurrent bouts of aspiration pneumonia.15
References
1. Atwater SW, Powers BE, Park RD, et al. Thymoma in dogs: 23 cases (1980-1991). J Am Vet Med Assoc 1994;205:1007-1013.
2. Messinger JS, Windham WR, Ward CR. Ionized hypercalcemia in dogs: a retrospective study of 109 cases (1998-2003). J Vet Intern Med 2009;23(3):514-519.
3. Rottenberg S, Von Tscharner C, Roosje PJ. Thymoma-associated exfoliative dermatitis in cats. Vet Path 2004;41(4):429-433.
4. Tepper LC, Spiegel IB, Davis GJ. Diagnosis of erythema multiforme associated with thymoma in a dog and treated with thymectomy. J Am Anim Hosp Assoc 2011;47(2):19-25.
5. Barton AD. T-cell lymphocytosis associated with lymphocyte-rich thymoma. Cancer 1997;80(8):1409-1417.
6. Burton AG, Borjesson DL, Vernau W. Thymoma-associated lymphocytosis in a dog. Vet Clin Path 2014;43(4):584-588.
7. Pentz WH. Advanced heart block as a manifestation of a paraneoplastic syndrome from malignant thymoma. Chest 1999;116(4):1135-1136.
8. Zitz JC, Birchard SJ, Couto GC, et al. Results of excision of thymoma in cats and dogs: 20 cases (1984-2005). J Am Vet Med Assoc 2008;232:1186-1192.
9. Day MJ. Review of thymic pathology in 30 cats and 36 dogs. J Small Anim Pract 1997;38(9):393-403.
10. Vail DM, Page RL. Miscellaneous tumors. In: Withrow & MacEwen's small animal clinical oncology. 5th ed. St. Louis: Elsevier-Saunders, 2013.
11. Aronsohn MG, Schunk KL, Carpenter J, et al. Clinical and pathologic features of thymoma in 15 dogs. J Am Vet Med Assoc 1984;184(11):1355-1362.
12. Shelton GD, Schule A, Kass PH. Risk factors for acquired myasthenia gravis in dogs: 1,154 cases (1991-1995). J Am Vet Med Assoc 1997;211(11):1428-1431.
13. Garneau MS, Price LL, Withrow SJ, et al. Perioperative mortality and long?term survival in 80 dogs and 32 cats undergoing excision of thymic epithelial tumors. Vet Surg 2015;44(5):557-564.
14. Hague DW, Humphries HD, Mitchell MA, et al. Risk factors and outcomes in cats with acquired myasthenia gravis (2001-2012). J Vet Intern Med 2015;29(5):1307-1312.
15. Shelton GD. Myasthenia gravis and disorders of neuromuscular transmission. Vet Clin North Am Small Anim Pract 2002;32(1):189-206.
16. Shelton GD. Myasthenia gravis: lessons from the past 10 years. J Small Anim Pract 1998;39:368-372.
17. Ettinger SJ, Feldman EC. Disorders of the esophagus. In: Textbook of veterinary internal medicine. 8th ed. Philadelphia; Elsevier health sciences, 2017.
18. Hall JA, Washabau RJ. Gastrointestinal prokinetic therapy: dopaminergic antagonist drugs. Compend Contin Ed Pract Vet 1997;19:214-221.
19. Lappin MR, Blondeau J, Boothe D, et al. Antimicrobial use guidelines for treatment of respiratory tract disease in dogs and cats: antimicrobial guidelines working group of the International Society for Companion Animal Infectious Diseases. J Vet Intern Med 2017; 31(2): 279-294.
Newsletter
From exam room tips to practice management insights, get trusted veterinary news delivered straight to your inbox—subscribe to dvm360.