Canine and feline nasal tumors
Detecting this mainly localized disease process early is difficult but important to improve an animal's prognosis.
Primary tumors of the nasal cavity account for about 1% or 2% of all neoplasms in dogs.1,2 Of these neoplasms, about 80% have histologic characteristics of malignancy.1 In dogs, nasal neoplasms are regionally invasive with frequent expansile growth into the nasal passages, frontal sinuses, and cranial vault cavity; however, documented distant metastasis to the lungs or regional draining lymph nodes is uncommon upon initial presentation.3,4
Carcinomas are more common than sarcomas and account for 60% to 78% of all nasal tumors in dogs.1,3-5 In five studies analyzing 711 nasal tumors in dogs, adenocarcinoma was the most common histologic subtype (45%), followed by squamous cell carcinoma (20%), chondrosarcoma (14%), undifferentiated or anaplastic carcinoma (11%), and unspecified carcinoma (10%).1,3,4,6,7

Getty Images
Nasal tumors are less commonly diagnosed in cats than in dogs but, nonetheless, behave similarly to canine nasal tumors with regional invasion and a low metastatic rate.1 Lymphoma and carcinoma (adenocarcinoma, squamous cell carcinoma) are the most common types of nasal tumor diagnosed in cats.1,8-10
Other less frequently diagnosed nasal malignancies that have been reported in dogs and cats include fibrosarcoma, mast cell tumor, transmissible venereal tumor, hemangiosarcoma, neuroendocrine carcinoma (olfactory neuroblastoma), peripheral nerve sheath tumor, fibrous histiocytoma, rhabdomyosarcoma, leiomyosarcoma, and melanoma.1,3,4 Benign tumors may also arise from the nasal cavity, including nasal polyps and fibromas, but they are not discussed in this review article.
SIGNALMENT
Age
Similar to other neoplastic conditions that predominantly arise in the geriatric population, nasal tumors most commonly affect middle-aged to older dogs and cats. The average age of onset for nasal carcinomas is reported to be 9 or 10 years, while the development of nasal sarcomas is reported to occur in slightly younger animals (7 or 8 years old).3,4,9,11-16
Breed
Although nasal tumors may be diagnosed in any dog breed, dolichocephalic and mesocephalic breeds are considered to be at an increased risk for developing nasal tumors.15-18 One potential hypothesis for this observed breed predilection is that the greater nasal passage surface area in dolichocephalic and mesocephalic dogs predisposes them to greater exposure to inhaled carcinogens and subsequent tumor development. In addition, the larger nasal surface area would be associated with a greater number of nasal epithelial cells, which could be malignantly transformed after exposure to inhaled carcinogens.
Sex
Reports vary on whether there is a male sex predilection for nasal tumors in dogs. As such, it is unlikely that the development of nasal tumors is under hormonal influence. Contrary to dogs, male cats have been reported to have about a twofold increased risk for developing nasal tumors, although an underlying mechanism to explain this observation has yet to be postulated.1
ETIOLOGY AND RISK FACTORS
Environmental carcinogens
Urban environments, with consequential nasal filtering of pollutants and exposure to environmental tobacco smoke, have been suggested to increase the risk of nasal tumor development.17,19 Although biologically reasonable, this possible influence still remains debatable since there are too few studies to draw definitive conclusions.
p53 expression
Known as the "guardian of the genome," the p53 gene produces p53 protein, which is essential for two key cellular processes—apoptosis and cell cycle regulation. The loss of p53 gene function within a cell predisposes it to genomic instability and predilection for acquiring gene mutations, which may lead to malignant transformation. Mutations in the p53 gene are frequently identified in a variety of tumor histopathologies, and evidence suggests that a mutated p53 gene may increase the risk of developing nasal tumors in dogs. Wild type (nonmutated) p53 protein has a short half-life and, thus, is typically not identified in a large proportion of normal cells. However, mutant p53 protein has an extended half-life secondary to protein conformational changes, and its accumulation and identification within the nucleus of malignant cells suggests that p53 protein may be involved in malignant transformation. In one study, the accumulation of p53 protein in the nucleus by immunohistochemistry was detected in 11 of 19 nasal adenocarcinomas, which suggests that p53 gene and protein mutation may participate in nasal tumorigenesis.20
COX-2 expression
Inflammatory eicosanoids, including prostaglandins and thromboxanes, participate in many physiologic and pathologic processes. Two enzymes, cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2), are responsible for the production of eicosanoids. COX-2 activity is induced by inflammatory stimuli and has been associated with tumorigenesis. Three studies evaluating COX-2 expression in canine nasal tumors found that 71% to 87% of nasal carcinomas are COX-2 positive,12,21,22 suggesting that COX-2 activity may participate in nasal tumorigenesis through suppression of apoptosis, promotion of angiogenesis and tumor invasion, and stimulation of cell proliferation.12
BIOLOGIC BEHAVIOR
Nasal tumors are characterized by rapid and progressive local tissue invasion but a low metastatic rate. Humane euthanasia of dogs and cats with nasal tumors is usually the result of local tumor progression rather than the development of metastatic disease. Varying between histopathologic types and investigational studies, the metastatic rate reported for nasal tumors in dogs ranges from 0% to 28% from the time of presentation to death.1,3,4,11,16,18,23
One recent study reported a metastatic rate of 8%, with identified metastases limited to the local lymph nodes, agreeing with other historical studies reporting a low incidence of pulmonary metastases in dogs.16,18 Although the incidence of regional and distant metastases for nasal tumors is relatively low, the histopathologic type may influence metastatic behavior. In one study based on histologic findings at necropsy, dogs with nasal carcinomas had a higher rate of metastases than dogs with nasal sarcomas had, suggesting a more aggressive metastatic phenotype for these epithelial-type tumors.3
Even within epithelial-type tumors, carcinomas may be subcategorized as being less or more aggressive. Dogs with aggressive carcinomas, such as anaplastic carcinoma or squamous cell carcinoma, generally have shorter survival times than dogs with less aggressive carcinomas, including adenocarcinoma and well-differentiated carcinoma. The median survival time of dogs with aggressive carcinomas and less aggressive carcinomas has been reported to be 7.2 and 11.9 months, respectively, compared with 24.1 months for dogs with sarcomas.6
CLINICAL SIGNS
The most common clinical signs seen in animals with nasal tumors include epistaxis, facial asymmetry, nonhemorrhagic nasal discharge, and sneezing. Physical examination findings may include stertorous breathing, enlarged mandibular lymph nodes, neurologic signs, decreased retropulsion of the eyes, exophthalmos, ocular discharge resulting from nasolacrimal duct obstruction, and overt facial deformation (Figure 1).

Figure 1. A mixed-breed dog with nasal carcinoma with direct extension of disease through the caudal nasal sinuses and into overlaying soft tissue. (Image courtesy of Louis-Philippe de Lorimier, DVM, DACVIM [oncology].)
Although a facial deformity is highly suggestive of a cancerous process, other differential diagnoses should include fungal or bacterial rhinitis, a foreign body, trauma, developmental abnormalities, and dental pathology. Systemic clinical signs including inappetence, lethargy, and weight loss may also be reported.13,16,23,24
Epistaxis is a common clinical sign in dogs and cats with nasal tumors. Most dogs (about 85%) with nasal neoplasia will manifest with frank hemorrhagic or serosanguinous nasal discharge, which correlates with a poorer prognosis.13,18,25 Although common in dogs with nasal cancer, epistaxis is not pathognomonic and is frequently observed in dogs with nonneoplastic rhinitis.18,25,26 In one retrospective study with 35 dogs presenting for evaluation of epistaxis, the underlying cause of nasal hemorrhage in 19 dogs (54%) was secondary to neoplasia.27
DIAGNOSIS
A thorough history and complete physical examination are the first step in assessing patients that may have nasal cancer. The history should include questions regarding the duration and progression of the clinical signs. A minimum database including a complete blood count, serum chemistry profile, and urinalysis is recommended to detect concurrent diseases. Systemic hypertension should be ruled out as a cause of epistaxis. A coagulation profile including prothrombin time, activated partial thromboplastin time, and bleeding time or activated clotting time is also recommended to rule out any coagulopathies before invasive diagnostic procedures.
Once a nasal mass is suspected or has been identified, regional lymph node aspiration, three-view thoracic radiography, imaging of the nasal cavity, and mass biopsy (rhinoscopy-assisted or blind) should be considered to make a definitive diagnosis. Although conventional radiography may prove to be beneficial, especially with open-mouth views for identifying space-occupying lesions within the nasal or frontal sinuses, computed tomography (CT) or magnetic resonance imaging (MRI) of the nasal cavities better assesses the extent of pathology before biopsy, surgery, or radiation therapy.25,28
Skull radiography
Skull radiography may be used as an initial diagnostic test to evaluate for bone lysis or soft tissue opacities within the nasal cavities, which may be suggestive of neoplasia. However, given the anatomic complexity of the canine and feline nasal cavities and the overlapping bony structures, definitive interpretation of routine radiographs may be diagnostically challenging as well as insensitive for assessing the extent of pathology. In a retrospective study of 42 dogs presenting for nasal disease, only 10% of the inflammatory rhinitis cases and 64% of the neoplastic cases were accurately diagnosed using conventional radiography alone.18 The lack of sensitivity and specificity associated with routine radiography, in conjunction with medical imaging advancements, has increased the use of advanced imaging modalities for documenting nasal cavity pathologies.
Advanced imaging
In general, CT provides superior imaging and visualization of bone, while MRI has greater resolution of soft tissue structures.29,30 Although CT or MRI may theoretically be better suited for identifying disease in specific clinical scenarios, in one small study of eight dogs with nasal tumors, no difference was seen between CT and MRI for the detection of intracalvarial changes, suggesting that either modality is appropriate for the identification of nasal tumors.29
However, given the fact that most nasal tumors involve bony structures including nasal turbinates and sinuses, CT is more commonly used to assess the extent of nasal disease as well as to differentiate between neoplastic and nonneoplastic diseases.
With CT, nasal tumors are typically characterized by extensive unilateral turbinate destruction and are often accompanied by erosive pathology involving contralateral turbinates, nasal septum, frontal sinuses, and the cribriform plate (Figure 2).25,28 In addition to identifying osteolytic abnormalities, CT imaging can also delineate soft tissue disease extension, such as involvement of the nasopharynx and periorbital regions.25 Finally, CT is integral for defining tumor margins, which is necessary before instituting curative-intent radiation therapy or en bloc surgical resection.
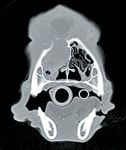
Figure 2. Computed tomography provides high-detail images of a tumor-infiltrated nasal cavity. Note the soft tissue mass growing within the nasal passage with concurrent osteolytic destruction and the extension of soft tissue growth dorsal to the nasal cavity.
Cytology or biopsy
Any suspect nasal mass effect may be aspirated to provide a rapid preliminary cytologic diagnosis. However, fine-needle aspiration is not always anatomically feasible because most nasal tumors are relatively inaccessible because of their location within the nasal cavity. Occasionally, nasal tumors associated with marked bony lysis of the nasal bone, frontal sinus, or maxilla may allow for direct disease extension outwards of the nasal cavity, resulting in a subcutaneous mass effect that may be sampled easily with fine-needle aspiration.
More commonly, a definitive diagnosis of nasal neoplasia is achieved through biopsy techniques, which are considered to be safe and only rarely induce unacceptable morbidity.31
Biopsy samples may be obtained by a variety of methods including
1. A Baker's punch biopsy of superficial facial deformities
2. Blind intranostril sample collection by using cup forceps or a bone curette
3. A rhinoscopy-assisted biopsy
4. An otoscopic illuminator-assisted biopsy of rostral lesions
5. An open-rhinotomy biopsy
6. Aggressive nasal flushing with subsequent dislodgement of tumor fragments
Before biopsy, it is recommended to perform a CT scan to maximize the likelihood of obtaining a representative tissue sample for definitive diagnosis. Although blind-biopsy techniques preclude direct visualization of suspect tumor masses, the relatively narrow confines of the nasal cavity afford good chances for the retrieval of tissue samples of diagnostic quality. Relatively noninvasive, direct-visualization techniques, such as rhinoscopy-assisted biopsy or otoscopic illuminator-assisted biopsy, are also commonly used to obtain a definitive diagnosis; however, they may be limited to retrieving small and superficial pieces of mass lesions.
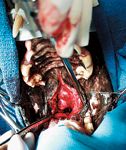
Figure 3. A ventral rhinotomy approach through the soft palate allows for the visualization and exposure of a relatively small nasal carcinoma arising from the ventral meatus. (Image courtesy of Louis-Philippe de Lorimier, DVM, DACVIM [oncology].)
When nondiagnostic specimens are obtained by using noninvasive techniques, a surgical technique may be required for definitive diagnosis. Surgical approaches for obtaining diagnostic samples include either a dorsal or ventral approach to the nasal cavity or an approach to the nasopharynx through the soft palate (Figure 3). If a surgical approach is required to obtain a biopsy, a CT scan—if not already performed—is highly recommended for optimal surgical planning.
STAGING
Staging of nasal tumors can be addressed through categorizing local invasiveness (Tables 1 & 2) and metastatic behavior (Table 2). Three-view thoracic radiography and fine-needle aspiration of regional lymph nodes will provide the information necessary to determine the stage based on metastatic behavior. A CT scan will provide highly valuable information regarding local invasiveness in staging and planning for surgery or radiation therapy. Fine-needle aspiration and cytologic examination of regional lymph nodes provide not only a rapid screening test for the presence of regional metastasis but may also allow for histopathologic categorization of the primary tumor (epithelial, mesenchymal, or other). The mandibular and retropharyngeal lymph nodes should be palpated and aspirated to check for regional metastases. Irrespective of lymph node size or lateralization, cytologic examination of aspirates from all draining lymph nodes should be performed to assess for micrometastases.

Table 1: Adams Modified Staging System for Nasal Tumors*
In addition to lymph node assessment, consider three-view thoracic radiography to rule out the existence of distant metastasis. At the time of nasal tumor diagnosis, radiographic findings in most dogs are usually unremarkable since the metastatic rate for nasal tumors is low. However, thoracic radiography may still be recommended since the prognosis and treatment planning will change if metastatic lung nodules or concurrent cardiac or pulmonary disease are present. To exemplify the impact of clinical stage on prognosis, in one study of 64 dogs with nasal adenocarcinoma, patients initially presenting with confirmed regional (lymph node) or distant (pulmonary parenchyma) metastases had substantially shorter survival times (109 days) than dogs presenting with only localized disease had (393 days).11
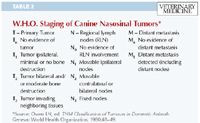
Table 2: W.H.O. Staging of Canine Nasosinal Tumors*
TREATMENT AND PROGNOSIS IN DOGS
Therapeutic options for dogs with nasal tumors are directed at local disease control. Given the restrictive anatomy of the nasal cavity and associated sinuses as well as the proximity to delicate tissues like the eyes and brain, complete surgical resection of nasal tumors is frequently difficult. Aggressive cytoreductive surgery also causes a high rate of morbidity with no significant improvement in survival times compared with dogs treated only with palliative medications.5,11,13,32,33 As such, surgery is not considered a first-line treatment option, and the standard of care is radiation therapy alone or a multimodality strategy combining radiation therapy with surgery or chemotherapy.
New advances in radiation delivery techniques that maximally spare normal tissue, such as intensity-modulated radiation therapy (IMRT),34 helical tomography,35 and proton therapy,36 are also being evaluated for improving treatment outcomes in dogs with nasal tumors. Despite advances in therapeutic strategies, anywhere from 63% to 100% of dogs treated with definitive radiation therapy protocols will experience local disease recurrence.37-42
Radiation therapy
The delivery of ionizing radiation with both orthovoltage or megavoltage therapy machines has been used for curative intent and palliative therapy for nasal tumors. Radiation therapy has the advantage of treating the entire nasal cavity, including bone, and its use has been associated with the greatest improvement in survival when compared with nonradiation treatment options. A notable limitation of radiation, when used as the sole therapeutic modality, is the low cure rate achieved in the treatment of large macroscopic tumor burdens. Despite radiation therapy's inability to cure most dogs, many patients treated with radiation therapy enjoy relatively long durations of local disease control, improved clinical signs, and increased quality of life scores.
Definitive treatment. Radiation therapy with curative intent has been previously described as a sole treatment option for nasal tumors in dogs. Conventional protocols require the administration of small fractions (3 to 4.2 Gy) repeatedly (10 to 19 treatments) on a daily or every other day basis for a total radiation dosage of 40 to 57 Gy.11,23,38,39 Three studies involving 82 dogs with nasal tumors that were treated with cobalt-60 radiation therapy alone reported median survival times ranging from 12 to 19.7 months.37,38,43 In these three studies, no significant differences in survival times were identified in dogs with either carcinomas or sarcomas. Interestingly, in a smaller pilot study, histopathologic subtype did appear to influence response to therapy. In this small descriptive study, six dogs with nonkeratinizing squamous cell carcinoma of the nasal cavity treated with cobalt-60 radiation therapy achieved only a median survival time of 165 days, suggesting that squamous cell carcinoma (Figure 4) may be a histopathologic subtype of nasal carcinoma that does not respond as well to radiation therapy.44
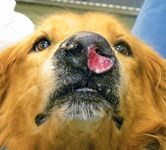
Figure 4. A nasal squamous cell carcinoma involving the rostral aspect of the left nasal cavity in a golden retriever. Note the large area of ulcerative tumor growth.
Definitive treatment side effects. Ionizing radiation exerts its therapeutic effects through the direct and indirect breakage of cellular DNA. Thus, tumor cells exposed to ionizing radiation will undergo programmed cell death as a consequence of irreparable DNA damage. This net effect of cell death translates clinically into tumor mass shrinkage. However, damage done to cellular DNA by ionizing radiation is not specific for tumor cells but will affect all cells within the field of radiation, including normal stromal cells associated with the nasal cavity. As such, therapy with ionizing radiation is often associated with adverse effects to surrounding normal tissues, which may be categorized as either acute or late effect radiation toxicities. Acute effects develop in tissues with rapidly dividing cells such as basal epithelial cells, and these radiation side effects are generally self-limiting. Tissues most commonly affected with acute effects include skin (desquamation), eyes (keratoconjunctivitis and blepharitis), and mucosal surfaces (mucositis). Acute radiation effects develop during the first three or four weeks of therapy and typically resolve within two or three weeks after treatment completion (Figures 5A & 5B).
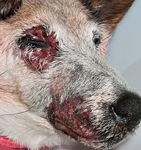
Figure 5A. In a mixed-breed dog receiving definitive radiation therapy for nasal carcinoma, painful acute radiation side effects can be observed involving the skin surrounding the eye and muzzle. (Image courtesy of Louis-Philippe de Lorimier, DVM, DACVIM [oncology].)
Although transient in nature, the management of acute effects is important and should include pain relief with nonsteroidal anti-inflammatory agents or opioids. Oral antibiotics and artificial tears may also be necessary for supportive care until the acute effects resolve completely. Late side effects of radiation therapy affect tissues with slower or minimal replicative capacity including bone, blood vessels, and neuronal tissue. Late side effects of radiation are dose-limiting and dictate maximum tolerated dosages of ionizing radiation deliverable to normal tissues within the treatment field. The onset of late radiation side effects occurs six months or longer after the completion of radiation therapy, and associated damage to normal tissues can be irreversible and associated with significant morbidity (Figure 6). In some cases, additional diagnostic testing may be necessary to differentiate between late radiation effects and tumor recurrence.

Figure 5B. Several months after the completion of definitive radiation therapy, the damage from acute radiation toxicity has healed, leaving regions of alopecia and hyperpigmentation. The right eye globe was included in the radiation field, and the dog did go on to develop mild cornea edema. (Image courtesy of Louis-Philippe de Lorimier, DVM, DACVIM [oncology].)
Given the potential for normal tissue damage during radiation therapy, recent technological advances have focused on increasing conformal targeting to minimize the volume of normal tissue exposed to the ionizing effects of radiation, with subsequent reductions in early and late radiation toxicity. Two technologies that allow for conformal therapy include stereotactic radiosurgery and IMRT. Two studies evaluating IMRT in dogs with nasal tumors have been reported. In the first study, IMRT was evaluated in 12 dogs with nasal tumors with the intent to document IMRT's therapeutic effectiveness as well as to characterize the frequency and severity of side effects.34 In these 12 dogs, the use of IMRT provided a median survival time of 14.9 months, with a low incidence of late effects.34 In a second larger study, IMRT was used to treat 31 dogs with sinonasal tumors with the intent of demonstrating an ocular-sparing effect in conjunction with anticancer activities.45 In this study, IMRT exerted similar anticancer effects as conventional two-dimensional radiation therapy (with survival times of 411 days vs. 420 days, respectively) but dramatically reduced the incidence and severity of early and late effects to the eyes.45 These two early IMRT studies indicate that this radiation delivery technique provides therapeutic effects equivalent to conventional two-dimensional radiation therapy but, more important, reduces the incidence and severity of radiation toxicity to surrounding normal tissues.
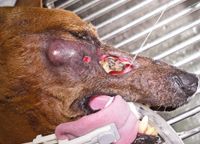
Figure 6. After treatment with large fraction palliative therapy, a dachshund developed unacceptable late radiation side effects manifested as osteonecrosis of the maxillary cavity. The open maxillary cavity with necrotic bone is being irrigated with a stream of sterile saline solution. It is probable that there is local tumor recurrence (mass ventral to the eye), but it was not confirmed in this case. The prognosis for the patient was very poor given that the late radiation effects were irreversible.
Radiation therapy with surgery. Some debate exists over combining radiation therapy with surgical resection for the management of canine nasal tumors. Most studies do not demonstrate any added benefit when surgery is combined with radiation therapy for the localized management of nasal tumors. In a recent analysis of 139 dogs with nasal tumors treated with radiation alone or radiation with cytoreductive surgery, median survival times were not significantly improved by the addition of cytoreductive surgery, which is consistent with many previous studies that also suggest that survival time remains unchanged with the addition of surgery to radiation therapy.4,7,23,24,41,46 Collectively, these studies indicate that surgical cytoreduction before the delivery of radiation is not beneficial for extending survival times.
While earlier studies evaluated the effects of surgery followed by radiation therapy, one recent study assessed the effects of radiation therapy followed by surgical exenteration of the nasal cavity. In this study, 53 dogs were treated with radiation therapy delivered in 10 fractions of 4.2 Gy, and in a small subset (n=13) of dogs, surgical exenteration of the nasal cavity was performed to remove any residual tumor burden. Interestingly, dogs adjuvantly treated with nasal cavity surgical exenteration had a median survival time of 47.7 months—much longer than dogs receiving only radiation therapy, which had a median survival time of 19.7 months.38 The one-, two-, and three-year survival rates for the radiation therapy-only group were 68%, 44%, and 24%, respectively. For the radiation therapy-and-surgical-exenteration group the survival rates were 77%, 69%, and 58%, respectively. Local tumor recurrence rates were not significantly different between treatment groups, but chronic complications including osteomyelitis and rhinitis were significantly higher in dogs treated with radiation therapy and surgical exenteration.
Radiation therapy combined with chemotherapy. Systemic chemotherapy has been classically indicated for the treatment of disseminated metastatic disease. However, the achievement of high local concentrations within the primary tumor microenvironment may allow for systemic chemotherapy to exert direct anticancer activities, which may contribute to the localized control of nasal tumors. Few studies have been conducted to determine the single-agent effectiveness of chemotherapy for the management of nasal tumors; however, some evidence would suggest that cisplatin in dogs exerts direct anticancer effects at the level of the primary nasal tumor.47
Despite some evidence for single-agent chemotherapy in the management of nasal tumors, a greater number of studies have focused on evaluating chemotherapeutics, such as cisplatin, as radiosensitizing agents rather than direct cytotoxic drugs. In a small preliminary study, 13 dogs with nasal tumors were treated with megavoltage radiation and a slow release polymer impregnated with cisplatin (implanted intramuscularly at a distant site) at a dosage of 60 mg/m2 to evaluate the feasibility and safety of this novel polymer system as a radiosensitizer. The release of cisplatin by the polymer system did not cause any systemic drug toxicosis; however, a low incidence (15%) of local tissue reaction at the site of polymer implantation was documented. More important, acute radiation side effects to normal tissue within the prescribed radiation field did not appear to be exacerbated by radiosensitization. Impressively, the median survival time achieved in this small preliminary study was 580 days, a clinically substantial improvement over historical radiation studies.48
In a larger follow-up study using the same combination protocol, 51 dogs were treated and also demonstrated unusually good survival times (median 15.8 months). Despite the positive survival times, most dogs (67%) still experienced disease relapse or progression.39 Similar to the original pilot study, it was confirmed that the cisplatin polymer was well-tolerated and did not seem to increase the incidence of early- or late-radiation toxicity effects.39
Finally, a third study compared the survival times between dogs with nasal tumors treated with radiation alone or radiation in conjunction with every-other-day, low-dose (7.5 mg/m2 ) intravenous cisplatin. Although low-dose cisplatin administered every other day resulted in some dogs developing azotemia, overall the protocol was well-tolerated but failed to improve survival times compared with dogs treated with radiation alone.49
Palliative radiation therapy. The goal of palliative radiation therapy is to reduce the tumor burden and improve quality of life. Most commonly, palliative radiation protocols deliver large fractions of radiation (6 to 8 Gy fractions) once or twice weekly for a total of four to six treatments. This palliative dosing strategy typically ameliorates clinical signs associated with disease but is insufficient to dramatically reduce tumor burden for prolonged time periods as compared with definitive treatment protocols.
In one study, 56 dogs with nasal tumors were treated weekly for four consecutive weeks with 9 Gy fractions. Most dogs (53/56) experienced improvements in clinical signs and minimal late radiation toxicity effects. The median survival time was 212 days, with reported one- and two-year survival rates of 45% and 15%, respectively.14
In a second retrospective study, the responses and survival times of 48 dogs were characterized after treatment with varying palliative radiation protocols. The cumulate radiation dosage administered ranged from 16 to 40 Gy, reflecting the diversity in treatment protocols instituted. Despite the lack of uniformity in treatment protocols, clinical signs in most dogs (66%) resolved completely after therapy for a median duration of 120 days, and the overall median survival time of dogs in this study was 146 days.40
Most recently, a study evaluating coarsely fractionated, palliative radiation therapy based on CT-based three-dimensional treatment planning in 38 dogs with nasal tumors has been reported. The therapeutic effects of three palliative protocols were studied: 1) 8 Gy fractions x four treatments, 2) 6 Gy fractions x five treatments, and 3) 3 Gy fractions x 10 treatments. All dogs treated were subjectively reported to have amelioration in their clinical signs after the completion of palliative therapy, and the median survival time reported for this group of 38 dogs was 10 months. Dogs with early stage disease (T1) intuitively had longer progression-free intervals (21.3 months) than dogs with more advanced disease (T2) did (8.5 months).50
Additional supportive measures
Dogs not receiving any form of definitive therapy (radiation, chemotherapy, surgery) tend to have relatively short median survival times ranging from 95 to 153 days.5,7,13 However, several options should be considered for improving the quality of life for dogs not receiving more conventional treatments. Based on their invasive phenotype, it is likely that dogs with nasal tumors experience discomfort and pain, so analgesics should be instituted to improve quality of life scores.
TREATMENT AND PROGNOSIS IN CATS
Compared with dogs, fewer studies document treatment options and outcomes for cats with nasal tumors. The most common tumor histopathologies that develop in the nasal passages of cats are lymphoma and carcinoma. Given the exquisite sensitivity of lymphoma to conventional radiation and chemotherapy, a definitive diagnosis in cats with obstructive nasal disease is necessary to provide owners with the most accurate prognostic and therapeutic recommendations.
Several recent studies have described the outcome of cats with nasal lymphoma after treatment with radiation alone, chemotherapy alone, or a combination of radiation and chemotherapy. In one study, 19 cats with nasal lymphoma treated with fractioned megavoltage radiation (median dosage of 42 Gy) and six months of systemic multiagent chemotherapy achieved a median survival time of 945 days and a three-year survival rate of 26%.51 In a larger, multi-institutional retrospective study, 97 cats with nasal lymphoma were evaluated for survival time after treatment with radiation, chemotherapy, or both. Regardless of treatment options, the median survival time of cats in this study was 536 days. No significant differences in survival times among cats receiving radiation alone (n=19), chemotherapy alone (n=18), or the radiation and chemotherapy combination (n=60) were detected, suggesting that nasal lymphoma may be equally and effectively managed with any of the evaluated treatment combinations.52 Finally, a recent study evaluated the responsiveness of feline extranodal lymphomas to systemic chemotherapy. Cats with nasal lymphoma achieving a complete response after chemotherapy had a median survival time of 749 days.53 Collectively, these studies indicate that feline nasal lymphoma responds well to conventional treatment options, including radiation and systemic chemotherapy (Figures 7A & 7B).
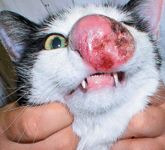
Figure 7A. Atypical feline nasal lymphoma, presenting with disfiguring tumor proliferation involving the apical aspect of the nasal planum. (Image courtesy of Louis-Philippe de Lorimier, DVM, DACVIM [oncology].)
Even though there are few studies characterizing nonlymphoproliferative feline nasal tumors, the prognosis in cats with such tumors appears to be shorter than cats with nasal lymphoma. In a small pilot study of eight cats with nonlymphoproliferative nasal tumors treated with megavoltage radiation therapy, the median survival time was 382 days, which is similar to the prognosis in dogs with radiation-treated nasal tumors.54

Figure 7B. The cat in Figure 7A after treatment with megavoltage radiation therapy. Remarkable complete response is achieved with near normalization of facial conformation. (Image courtesy of Louis-Philippe de Lorimier, DVM, DACVIM [oncology].)
SUMMARY
Nasal tumors in dogs and cats are usually a localized disease process, with low frequency of metastases. Early detection is often difficult because of the occult nature of nasal tumor development; however, timely therapeutic intervention is important to minimize extensive invasion into neighboring structures, including the central nervous system and orbital cavities. The standard of care for definitive treatment of nasal tumors confined to the nasal cavity in dogs and cats is radiation therapy alone or radiation therapy combined with surgery or chemotherapy. Although initially responsive to conventional radiation therapy allowing for long-term disease control, most dogs and cats eventually experience locally recurrent disease. With the greater availability of sophisticated radiation delivery units for use in veterinary medicine, including IMRT and stereotactic radiosurgery, it is likely that these advanced radiation delivery methods will improve management and treatment strategies for nasal tumors in dogs and cats in the near future.
Zachary L. Neumann, DVM*
Timothy M. Fan, DVM, PhD, DACVIM (internal medicine, oncology)
Department of Veterinary Clinical Medicine
College of Veterinary Medicine
University of Illinois
Urbana, IL 61802
Jayme Looper, DVM, DACVR (radiation oncology)
VCA Aurora Animal Hospital
2600 West Galena Blvd
Aurora, IL 60506
*Current address: Department of Clinical Sciences
College of Veterinary Medicine
University of Minnesota
St. Paul, MN 55108
REFERENCES
1. Madewell BR, Priester WA, Gillete EL, et al. Neoplasms of the nasal passages and paranasal sinuses in domesticated animals as reported by 13 veterinary colleges. Am J Vet Res 1976;37(7):851-856.
2. Brodey RS. Canine and feline neoplasia. Adv Vet Sci Comp Med 1970;14:309-354.
3. Patnaik AK. Canine sinonasal neoplasms: clinicopathological study of 285 Cases. J Am Anim Hosp Assoc 1989;25:103-114.
4. LaDue TA, Dodge R, Page RL, et al. Factors influencing survival after radiotherapy of nasal tumors in 130 dogs. Vet Radiol Ultrasound 1999;40(3):312-317.
5. MacEwen EG, Withrow SJ, Patnaik AK. Nasal tumors in the dog: retrospective evaluation of diagnosis, prognosis, and treatment. J Am Vet Med Assoc 1977;170(1):45-48.
6. Adams WM, Kleiter MM, Thrall DE, et al. Prognostic significance of tumor histology and computed tomographic staging for radiation treatment response of canine nasal tumors. Vet Radiol Ultrasound 2009;50(3):330-335.
7. Yoon JH, Feeney DA, Jessen CR, et al. External-beam Co-60 radiotherapy for canine nasal tumors: a comparison of survival by treatment protocol. Res Vet Sci 2008;84(1):140-149.
8. Henderson SM, Bradley K, Day MJ, et al. Investigation of nasal disease in the cat—a retrospective study of 77 cases. J Feline Med Surg 2004;6(4):245-257.
9. Legendre A, Krahwinekel D, Spaulding K. Feline nasal and paranasal sinus tumors. J Am Anim Hosp Assoc 1981;17:1038-1039.
10. Mukaratirwa S, van der Linde-Sipman JS, Gruys E. Feline nasal and paranasal sinus tumours: clinicopathological study, histomorphological description and diagnostic immunohistochemistry of 123 cases. J Feline Med Surg 2001;3(4):235-245.
11. Henry CJ, Brewer WG Jr, Tyler JW, et al. Survival in dogs with nasal adenocarcinoma: 64 cases (1981-1995). J Vet Intern Med 1998;12(6):436-439.
12. Kleiter M, Malarkey DE, Ruslander DE, et al. Expression of cyclooxygenase-2 in canine epithelial nasal tumors. Vet Radiol Ultrasound 2004;45(3):255-260.
13. Rassnick KM, Goldkamp CE, Erb HN, et al. Evaluation of factors associated with survival in dogs with untreated nasal carcinomas: 139 cases (1993-2003). J Am Vet Med Assoc 2006;229(3):401-406.
14. Mellanby RJ, Stevenon RK, Herrtage ME, et al. Long-term outcome of 56 dogs with nasal tumours treated with four doses of radiation at intervals of seven days. Vet Rec 2002;151(9):253-257.
15. Morris JS, Dunn KJ, Dobson JM, et al. Radiological assessment of severity of canine nasal tumours and relationship with survival. J Small Anim Pract 1996;37(1):1-6.
16. Avner A, Dobson JM, Sales JI, et al. Retrospective review of 50 canine nasal tumours evaluated by low-field magnetic resonance imaging. J Small Anim Pract 2008;49:233-239.
17. Reif JS, Bruns C, Lower KS. Cancer of the nasal cavity and paranasal sinuses and exposure to environmental tobacco smoke in pet dogs. Am J Epidemiol 1998;147(5):488-492.
18. Tasker S, Knottenbelt CM, Munro EA, et al. Aetiology and diagnosis of persistent nasal disease in the dog: a retrospective study of 42 cases. J Small Anim Pract 1999;40(10):473-478.
19. Bukowski JA, Wartenberg D, Goldschmidt M. Environmental causes for sinonasal cancers in pet dogs, and their usefulness as sentinels of indoor cancer risk. J Toxicol Environ Health A 1998;54(7):579-591.
20. Gamblin RM, Sagartz JE, Couto CG. Overexpression of p53 tumor suppressor protein in spontaneously arising neoplasms of dogs. Am J Vet Res 1997;58(8):857-863.
21. Borzacchiello G, Paciello O, Papparella S. Expression of cyclooxygenase-1 and -2 in canine nasal carcinomas. J Comp Pathol 2004;131(1):70-76.
22. Impellizeri JA, Esplin DG. Expression of cyclooxygenase-2 in canine nasal carcinomas. Vet J 2008;176(3):408-410.
23. Northrup NC, Etuw SM, Ruslander DM, et al. Retrospective study of orthovoltage radiation therapy for nasal tumors in 42 dogs. J Vet Intern Med 2001;15(3):183-189.
24. Evans SM, Goldschmidt M, McKee LJ, et al. Prognostic factors and survival after radiotherapy for intranasal neoplasms in dogs: 70 cases (1974-1985). J Am Vet Med Assoc 1989;194(10):1460-1463.
25. Lefebvre J, Kuehn NF, Wortinger A. Computed tomography as an aid in the diagnosis of chronic nasal disease in dogs. J Small Anim Pract 2005;46(6):280-285.
26. Windsor RC, Johnson LR, Herrgesell EJ, et al. Idiopathic lymphoplasmacytic rhinitis in dogs: 37 cases (1997-2002). J Am Vet Med Assoc 2004;224(12):1952-1957.
27. Strasser JL, Hawkins EC. Clinical features of epistaxis in dogs: a retrospective study of 35 cases (1999-2002). J Am Anim Hosp Assoc 2005;41(3):179-184.
28. Saunders JH, van Bree H, Gielen I, et al. Diagnostic value of computed tomography in dogs with chronic nasal disease. Vet Radiol Ultrasound 2003;44(4):409-413.
29. Dhaliwal RS, Kitchell BE, Losonsky JM, et al. Subjective evaluation of computed tomography and magnetic resonance imaging for detecting intracalvarial changes in canine nasal neoplasia. Intern J Appl Res Vet Med 2004;2(3):201-208.
30. Drees R, Forrest LJ, Chappell R. Comparison of computed tomography and magnetic resonance imaging for the evaluation of canine intranasal neoplasia. J Small Anim Pract 2009;50(7):334-340.
31. Lent SE, Hawkins EC. Evaluation of rhinoscopy and rhinoscopy-assisted mucosal biopsy in diagnosis of nasal disease in dogs: 119 cases (1985-1989). J Am Vet Med Assoc 1992;201(9):1425-1429.
32. Laing EJ, Binnington AG. Surgical therapy of canine nasal tumors: a retrospective study (1982-1986). Can Vet J 1988;29(10):809-813.
33. Holmberg DL, Fries C, Cockshutt J, et al. Ventral rhinotomy in the dog and cat. Vet Surg 1989;18(6):446-449.
34. Hunley DW, Mauldin GN, Shiomitsu K, et al. Clinical outcome in dogs with nasal tumors treated with intensity-modulated radiation therapy. Can Vet J 2010;51(3):293-300.
35. Gutierrez AN, Deveau M, Forrest LJ, et al. Radiobiological and treatment planning study of a simultaneously integrated boost for canine nasal tumors using helical tomotherapy. Vet Radiol Ultrasound 2007;48(6):594-602.
36. Kaser-Hotz B, Sumova A, Lomax A, et al. A comparison of normal tissue complication probability of brain for proton and photon therapy of canine nasal tumors. Vet Radiol Ultrasound 2002;43(5):480-486.
37. McEntee MC, Page RL, Heidner GL, et al. A retrospective study of 27 dogs with intranasal neoplasms treated with cobalt radiation. Vet Radiol 1991;32(3):135-139.
38. Adams WM, Bjorling DE, McAnulty JE, et al. Outcome of accelerated radiotherapy alone or accelerated radiotherapy followed by exenteration of the nasal cavity in dogs with intranasal neoplasia: 53 cases (1990-2002). J Am Vet Med Assoc 2005;227(6):936-941.
39. Lana SE, Dernell WS, Lafferty MH, et al. Use of radiation and a slow-release cisplatin formulation for treatment of canine nasal tumors. Vet Radiol Ultrasound 2004;45(6):577-581.
40. Gieger T, Rassnick K, Siegel S, et al. Palliation of clinical signs in 48 dogs with nasal carcinomas treated with coarse-fraction radiation therapy. J Am Anim Hosp Assoc 2008;44(3):116-123.
41. Theon AP, Madewell BR, Harb MF, et al. Megavoltage irradiation of neoplasms of the nasal and paranasal cavities in 77 dogs. J Am Vet Med Assoc 1993;202(9):1469-1475.
42. Morris JS, Dunn KJ, Dobson JM, et al. Effects of radiotherapy alone and surgery and radiotherapy on survival of dogs with nasal tumours. J Small Anim Pract 1994;35:573-576.
43. Adams WM, Miller PE, Vail DM, et al. An accelerated technique for irradiation of malignant canine nasal and paranasal sinus tumors. Vet Radiol Ultrasound 1998;39(5):475-481.
44. Correa SS, Mauldin GN, Mauldin GE, et al. Efficacy of cobalt-60 radiation therapy for the treatment of nasal cavity nonkeratinizing squamous cell carcinoma in the dog. J Am Anim Hosp Assoc 2003;39(1):86-89.
45. Lawrence JA, Forrest LJ, Turek MM, et al. Proof of principle of ocular sparing in dogs with sinonasal tumors treated with intensity-modulated radiation therapy. Vet Radiol Ultrasound 2010;51(5):561-570.
46. Adams WM, Withrow SJ, Walshaw R, et al. Radiotherapy of malignant nasal tumors in 67 dogs. J Am Vet Med Assoc 1987;191(3):311-315.
47. Hahn KA, Knapp DW, Richardson RC, et al. Clinical response of nasal adenocarcinoma to cisplatin chemotherapy in 11 dogs. J Am Vet Med Assoc 1992;200(3):355-357.
48. Lana SE, Dernell WS, LaRue SM, et al. Slow release cisplatin combined with radiation for the treatment of canine nasal tumors. Vet Radiol Ultrasound 1997;38(6):474-478.
49. Nadeau ME, Kitchell BE, Rooks RL, et al. Cobalt radiation with or without low-dose cisplatin for treatment of canine naso-sinus carcinomas. Vet Radiol Ultrasound 2004;45(4):362-367.
50. Buchholz J, Hagen R, Leo C, et al. 3D conformal radiation therapy for palliative treatment of canine nasal tumors. Vet Radiol Ultrasound 2009;50(6):679-683.
51. Sfiligoi G, Theon AP, Kent MS. Response of nineteen cats with nasal lymphoma to radiation therapy and chemotherapy. Vet Radiol Ultrasound 2007;48(4):388-393.
52. Haney SM, Beaver L, Turrel J, et al. Survival analysis of 97 cats with nasal lymphoma: a multi-institutional retrospective study (1986-2006). J Vet Intern Med 2009;23(2):287-294.
53. Taylor SS, Goodfellow MR, Browne WJ, et al. Feline extranodal lymphoma: response to chemotherapy and survival in 110 cats. J Small Anim Pract 2009;50(11):584-592.
54. Mellanby RJ, Herrtage ME, Dobson JM. Long-term outcome of eight cats with non-lymphoproliferative nasal tumours treated by megavoltage radiotherapy. J Feline Med Surg 2002;4(2):77-81.
Newsletter
From exam room tips to practice management insights, get trusted veterinary news delivered straight to your inbox—subscribe to dvm360.