Understanding pharmacokinetics in the clinical setting (Proceedings)
Clinical pharmacokinetics is concerned with the rational, safe, and effective use of drugs. It studies the factors that determine the time course of the plasma concentration of a drug and its variability.
Clinical pharmacokinetics is concerned with the rational, safe, and effective use of drugs. It studies the factors that determine the time course of the plasma concentration of a drug and its variability. The ideal goal is to tailor the drug and dosing regimen to the unique characteristics of each patient1. Pharmacokinetic information contained in the drug package insert can be an important tool when there is a need to adapt the dosage regimen to particular clinical scenarios (e.g. renal impairment, change in bacterial susceptibility, etc). Pharmacokinetic information is most useful when the range of doses (or concentrations) in which the drug exhibits linear pharmacokinetics is known. Linear pharmacokinetics implies direct proportionality between dose and exposure. For example, under linear kinetics, a 20 mg/kg dose provides two-fold the exposure (but not necessarily the pharmacological effect) of a 10 mg/kg dose. Some drugs exhibit linear pharmacokinetics within a wide range of doses and concentrations, while others do not. Therefore, when examining the pharmacokinetic information in the drug insert we can start by determining the PK linearity and range. Under nonlinear pharmacokinetics, there is no proportionality between dose and exposure, and therefore dose adjustment becomes a much more difficult task. The principles discussed by the author are based on the assumption of linear PK (also called first-order PK), which is by far the most common scenario.
Clinical pharmacodynamics is concerned with the rational, safe and effective use of drugs. It studies the relationship between drug exposure and pharmacological or toxic effect. Pharmacodynamic studies often allow defining exposure targets that are optimally related to the desired level of pharmacological effect (e.g. a range of AUC values that optimizes the probability of bacterial eradication during an infection)2. Important therapeutic questions that may arise during the course of drug administration include:
What should be the dose interval? Is compliance likely to be a problem? Are there any potential interaction with the patient's other drugs? What if the "normal average" dosing regimen is not appropriate for this patient? Should liver or renal impairment be taken into account? How do I tailor the dose to the changing bacterial susceptibility in my patient? How should I interpret TDM findings? When both pharmacokinetic and pharmacodynamic information are available, clinicians are better equipped to provide rational answers to these and other questions as well as to calculate adjusted dosage regimens. One of the primary sources of PK and PD information is the package insert itself.
Objectives Of The Presentation
1. To review the PK and PD information included in the drug label.
2. To discuss the practical application of PK and PD information contained on the label.
Relevant Pharmacokinetic Information In The Drug Package Insert
Fraction of Drug Unbound in Plasma (fu)
Free drug in plasma can diffuse to tissues and act on receptors to produce an effect. While drug displacement from plasma protein binding sites by a competing drug is almost always unimportant, protein binding may be significant in the interpretation of plasma concentrations of drugs (e.g. when interpreting the results of therapeutic drug monitoring for displaced drugs or drugs with nonlinear plasma protein binding). Protein binding is also important with antimicrobials. When adjusting the dosage regimen of antimicrobials based on PKPD indices of efficacy (e.g. AUC/MIC or T>MIC), the exposure parameters (Cp, or AUC) should be expressed in terms of unbound drug (e.g. Cpu = Cp × fu). Expressed in this way, PKPD indices of efficacy can be extrapolated across species (e.g. from mice to dogs).
Area under the Plasma Concentration-Time Curve (AUC) and Other Measures of Exposure
The AUC represents the total area contained between the concentration-time profile and the X and Y axis of a pharmacokinetic curve. For a single dose, the AUC represents the complete exposure of an animal to a drug, i.e. the exposure from time 0 to infinity. As such, AUC after single dose is usually represented as AUC0-inf. When multiple doses are administered at regular dose intervals (τ) (with τ < 3-4 half-lives), the plasma concentration of the drug accumulates until a plateau is reached. At plateau or steady-state, the peak and trough concentrations remain unchanged with each dose3. Assuming time- and dose-independent PK, the AUC over a dose interval at plateau (AUC0-τ) equals the AUC0-inf following a single dose. In the case of antimicrobials administered by multiple doses, PK-PD relationships are based on the unbound AUC0-24, or the area corresponding to a 24-hr dosing interval at steady-state4 (or the corresponding sum of areas if the drug is administered at shorter intervals).
Other measures of exposure that can be relevant for dosage adjustment include Cmax, or peak plasma concentration after single or multiple doses; Cpss, or concentration at steady-state after IV constant-rate infusion, Cpavgss, or average concentration at steady state after multiple doses with constant dose interval; and Cmin, or trough concentration at steady-state after multiple doses. These measures of exposure are generally related to the volume of distribution and/or systemic clearance of a drug, and therefore are amenable to become targets for drug adjustment, as long as their values are associated to a given probability of clinical outcome.
Under certain circumstances, these measures of exposure may need to be adjusted by the fraction of free drug reaching the target tissue. In those cases, information on drug tissue distribution may allow the corresponding adjustment to be made.
Systemic Availability (F)
When a drug is administered by an extra-vascular route, a fraction of the administered dose may not reach the systemic circulation. This may be due to poor absorption, drug inactivation in the stomach, or enzymatic first-pass effect (in the intestinal wall or the liver). The bioavailability of a drug is formally defined as the rate and extent of absorption. Although the rate of absorption can be important when quick onset of pharmacological effects is needed, it is not usually relevant for long-term drug administration. The extent of absorption, known as systemic availability, represents the fraction of the total dose that reaches the systemic circulation after extravascular administration (usually PO, SQ, or IM) and may have a value between 0 and 1. Absolute systemic availability is determined by comparison between the dose-adjusted AUCs after IV and extravascular administrations, while relative systemic availability refers to the same calculation but for two extravascular administrations (e.g. PO vs IM):
Food may affect absorption and first-pass metabolism, typically in opposite ways. In monogastric species, food usually decreases the oral availability of drugs that are not very lipid soluble, while it tends to increase the oral availability of drugs that undergo high first-pass metabolism. Likewise, food components may specifically bind to certain drugs (e.g. calcium and tetracyclines) altering the absorption profile of the latter. While food effects are typically managed by appropriately timing the dose administration relative to the feeding process, dose adjustments can be made by applying an appropriate correcting factor to the value of F.
Systemic Clearance (Cl)
In first order pharmacokinetic systems, the clearance of a drug from plasma is a proportionality constant that relates the rate of elimination (mass/time) to the total plasma concentration (mass/volume). Systemic clearance is defined as the volume of plasma cleared of drug per unit time and is expressed in units of flow (volume/time). Clearance is the single most important pharmacokinetic parameter, since it determines the maintenance dose-rate (dose/unit time) required to maintain a given level of drug exposure. For example, for a drug administered intravascularly, the AUC is solely dependent on the dose and the drug systemic clearance. For a drug administered extravascularly, the factors affecting exposure are the dose, F, and Cl.
Therefore, to calculate the dose for a given target AUC and F, we only need to know the value of clearance:
When the overall elimination profile of a drug obeys linear kinetics, systemic clearance is considered constant and independent of the dose over the clinically useful range of plasma concentrations. If the eliminating organ is significantly impaired or a concurrently administered drug affects the clearance of the original drug, the dose or dose interval may need to be changed in proportion to the change in clearance.
Different animals treated with similar doses will exhibit different AUCs on account of the biological variability in clearance and systemic availability. Therefore, for a given value of F, it will be difficult to predict the individual AUC of drugs with very variable clearance5. Although a complete discussion on pharmacokinetic variability is beyond the scope of this lecture, it is important to recognize its influence on drug exposure predictions and dose calculations. As a general rule (assuming normal distribution), the AUC of drugs with coefficients of variation (standard deviation/average value) in clearance above 50% may be hard to predict in the individual patient. This may be of particular importance with critical patients, antimicrobials, and narrow therapeutic index drugs.
Other measures of exposure are linearly related to the systemic clearance as follows:
Therefore, for a given target Cpss
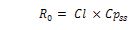
Where R0 is the zero-order rate of IV infusion.
When multiple doses are administered at constant dose intervals the average concentration at steady state (Cpavgss) is a function of the dose rate, F, and clearance in the same way as described above.
Volume of Distribution (Vd)
This is the second most important pharmacokinetic parameter for dose determination and adjustment. The volume of distribution is a function of the lipid solubility and plasma and tissue protein binding of the drug. It represents the volume into which a drug appears to be distributed with a concentration similar to that of plasma. This parameter is useful for determining the loading dose, i.e. the dose required to achieve the target plasma concentration (e.g. Cpavgss) in as little time as possible. This calculation may be important for the initial treatment of drugs with long half-lives (e.g. phenobarbital), since 4 half-lives are needed to reach concentrations in plasma above 90% of the target average concentration at steady state. For drugs that can be approximated with monocompartmental kinetic equations, the loading dose can be computed as follows:
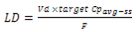
Elimination Half-Life (t½)
The terminal elimination half-life is the time that it takes for the concentration of the drug in plasma at any time to decrease by one-half. When the amounts of drug in plasma and tissue are in equilibrium, the half-life may also represent the time that it takes for the total amount of drug in the body to decrease by one-half. This parameter provides an indication of:
The time-course of drug elimination (by 4 half-lives, the plasma concentration of the drug will have decreased by about 93%).
The time-course of drug accumulation (when the dose interval is short enough relative to the half-life, it takes 4 half-lives to reach about 93% of the steady-state concentration).
The required dose interval, in particular for drugs with pharmacodynamic effects paralleling plasma concentrations. The choice of dose interval is also affected by the therapeutic index of the drug and the expected level of compliance. Half-lives that allow for once or twice-a-day administration are usually associated to increased compliance, in comparison to more frequent administration. Notice that since the half-life is a hybrid parameter of Vd and Cl:
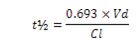
If Cl decreases (e.g. because of renal dysfunction), it may be possible to increase the dose interval (e.g. from 8 to 12 hr) increasing the chances for adequate compliance.
Pkpd-Based Dosage Regimen Design For Antibacterial Drugs
Outcome of infections in animal models has been shown to correlate well with one of three PKPD parameters, namely, the ratio of peak plasma concentration of the drug to the minimum inhibitory concentration (Cmax/MIC), the ratio of the 24-h area under the curve to the minimum inhibitory concentration (AUC/MIC), or the fraction of the dose interval during which the concentration of the drug in plasma remains above the minimum inhibitory concentration (T>MIC)6,7.
Pharmacokinetic (Cl, fu, Cmax, PK linearity range, etc) and bacterial susceptibility information (MIC50, MIC90) included in the package insert can be integrated with published information on well established PKPD targets and the bacterial susceptibility profile of a specific case in order to tailor the dosage regimen of antibiotics to the observed level of bacterial susceptibility.
Several examples will be presented during the lecture illustrating the use of pharmacokinetic information from the package insert to make rational decisions about dosage regimens.
References
Rodman JH, et al. Applied Pharmacokinetics and Pharmacodynamics, 4th ed. 2006; Burton ME, Shaw LM, Schentag JJ, Evans WE: Lippincott Williams & Wilkins, pp. 40-59.
Martin-Jimenez T. Farmacologia y Terapeutica Veterinaria 1st ed. 2002; Botana LM, Landoni F, Martin-Jimenez T: McGraw-Hill Interamericana, pp. 58-77.
Winter ME. Basic Clinical Pharmacokinetics, 4th ed. 2004; Winter ME: Lippincott Williams & Wilkins, pp.2-111.
Mouton JW, et al. J Antimicrob Chemother 2005;55:601.
Martin-Jimenez T, et al. Comparative Pharmacokinetics. Principes, Techniques and Applications, 1st ed. 1999; Riviere JE: Iowa State University Press, pp. 259-282.
Andes D, et al. Antibiotic Optimization. Concepts and Strategies in Clinical Practice, 1st ed. 2005; Owens R, Ambrose PG, Nightingale CH: Taylor and Francis, pp. 65-88.
Martinez M, et al. Antimicrobial Therapy in Veterinary Medicine, 4th ed. 2006; Giguere S, Prescott JF, Baggot JD, Walker RD, Dowling PM: Blackwell Publishing; pp. 81-106.
Key words: pharmacokinetics, pharmacodynamics, insert, antibacterials, label.