Salivation abnormalities of neurogenic origin-yet another reason to perform an oral examination
Max presented with unilateral temporalis muscle atrophy but also with changes to his saliva. Lets investigate what's behind it.
It's Monday morning. You walk into the office, back from a long weekend. You run through the list of the day's appointments and see that one of your favorite patients, Max, is on the day's schedule with the chief complaint being that the “head looks funny.”
Later that day, you enter the exam room and ask, “What's going on with Max today?”
His owner replies, “Well, doc, the left side of his head completely sunk in overnight!” You take one look at Max's head and your heart sinks-you quickly recognize that the left temporalis and masseter muscles have atrophied completely. Without the muscles, the left zygomatic arch is clearly visible to the naked eye. The left eye also has become enophthalmic with an accumulation of mucoid discharge and third eyelid elevation (Figure 1).
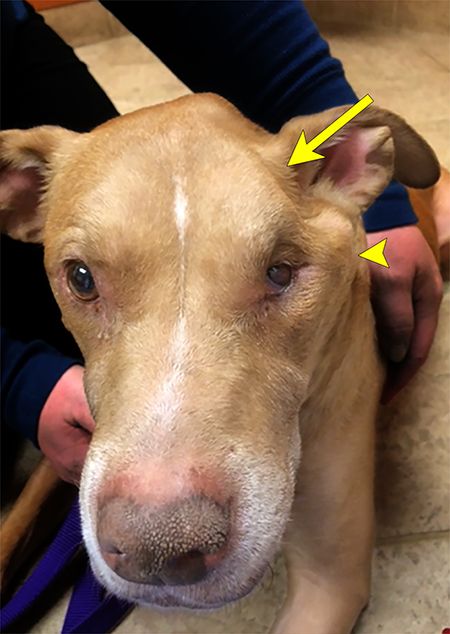
Figure 1. Max has unilateral dysfunction of the mandibular branch of cranial nerve V (trigeminal). Atrophy of the temporalis muscle (large arrow) is evident. Loss of the masseter and temporalis muscles makes the zygomatic arch more clearly defined (arrowhead). Additionally, enophthalmos of the left eye and secondary third eyelid elevation have resulted from atrophy of the pterygoid muscles. (All photos courtesy of Drs. Rachel B. Song, Eric N. Glass and Marc Kent)When you turn Max's head to examine the right side, you see how well-muscled his head is on the unaffected side. He's still able to close his eyelids when you perform the palpebral reflex and the menace response. You remember that to close the eyelids, cranial nerve VII (the facial nerve) has to be functioning normally. Likewise, all the other cranial nerves appear to have normal function on your examination. When you do an oral exam on Max, you see an accumulation of thick, foamy saliva in the caudal oropharynx only on the side of the muscle atrophy. You are puzzled and take a second and more thorough look to confirm your findings. Why is there foamy saliva in the back of his mouth, you wonder?
Thinking through the causes of muscle atrophy-disuse versus neurogenic atrophy-you think that it must be neurogenic atrophy, given how quickly the owner reported the onset of the atrophy. But then which nerve? Ah, but of course! You remember your vet school neurology training and recall that cranial nerve V, the trigeminal nerve, innervates the muscles of mastication. All those mnemonics you memorized to learn the cranial nerves and their functions are paying off. But then how to account for the abnormal saliva accumulation in Max's oropharynx?
A quick trigeminal nerve review
The trigeminal nerve has three main branches-the ophthalmic, maxillary and mandibular nerves. All three branches relay sensory information from the head and face to the brain.1 Only the mandibular branch is responsible for providing motor function to the muscles of mastication-the masseter, temporalis, lateral and medial pterygoids, rostral portion of the digastricus, and mylohyoideus muscles-as well as some lesser-known muscles such as the tensor tympani muscle (involved in modulating the ossicles in the middle ear) and the tensor veli palatani muscle (involved in opening the pharyngeal orifice of the auditory tube).
Clinically, unilateral dysfunction of the mandibular branch of the trigeminal nerve is easy to recognize, often because of the dramatic muscle atrophy of the masseter and temporalis muscles. Pterygoid muscle atrophy is inferred by the enophthalmos, as the pterygoid muscles provide ventral and medial support of the eye's position in the orbit. The third eyelid will elevate passively with the enophthalmia.
The salivary link
What probably was not a focus in your veterinary curriculum was the role that the trigeminal nerve serves in the autonomic nervous system. Branches of the trigeminal nerve act as a conduit for the distribution of parasympathetic innervation to target organs such as the lacrimal and salivary glands.
Anyone who has suffered from dry eye (keratoconjunctivitis sicca, or KCS), dry mouth (xerostomia) or dry nose (xeromycteria) can understand the life-altering effects such conditions can cause. Since veterinary patients can't self-report symptoms, most owners don't recognize altered autonomic function until severe end-stage consequences are present. To appreciate this, one only need examine a dog with untreated KCS. The lack of tear production leads to corneal opacification from corneal edema, neovascularization, pigment deposits, squamous metaplasia and hyperkeratinization of the cornea, which results in visual deficits.2 People with xerostomia suffer from severe oral discomfort, dental caries, speech problems and difficulty eating and swallowing.3
In dogs, there are four major salivary glands-the parotid, zygomatic, sublingual and submandibular glands.4 Although structural diseases of the salivary glands (i.e. sialocele, sialadenitis, salivary gland neoplasms and sialadenosis) are well-known in veterinary medicine, pure functional disturbances of the salivary glands have yet to be well-described.
Each of the salivary glands is innervated by the sympathetic and parasympathetic nervous system. As a whole, the autonomic nervous system has both afferent (sensory) and efferent (motor) components. Efferents are composed of a two-neuron system-preganglionic and ganglionic neurons. Sometimes these are referred to as first-order and second-order neurons, respectively. The preganglionic neuron is located in the central nervous system. Preganglionic axons synapse with the ganglionic neurons in various ganglia. Postganglionic axons innervate target organs such as glands, the heart, the lungs and many other organs.
For sympathetic innervation, preganglionic neurons are located in the intermediate gray matter of the thoracic and cranial lumbar spinal cord. For most of the body, the ganglionic neurons are located in the paired sympathetic trunk, which runs bilaterally along the ventrolateral aspect of the vertebral column. For structures of the head, ganglionic neurons are located in the cranial cervical ganglia near the ventral and caudal aspect of the skull. Long postganglionic axons course to their target organs. Sympathetic postganglionic axons use norepinephrine as their neurotransmitter.
For parasympathetic innervation, preganglionic neurons are located in the brain stem, adjacent to the motor neurons for cranial nerves III (oculomotor), VII (facial), XI (glossopharyngeal) and X (vagus), as well as sacral spinal cord segments. The ganglionic neurons are often located in ganglia close to their intended target organs. Short parasympathetic postganglionic axons course to their target organs and use acetylcholine as their neurotransmitter.
For the salivary glands, cranial nerves VII (facial) and IX (glossopharyngeal) provide for parasympathetic innervation.5 Specifically, preganglionic neurons for the parasympathetic innervation of the zygomatic and parotid glands are provided by the parasympathetic nuclei of the glossopharyngeal nerve in the medulla.5 Preganglionic axons travel in the glossopharyngeal nerve and synapse at the otic ganglion.5 The postganglionic axons course with the auriculotemporal nerve, a branch of the mandibular nerve, to arrive at the zygomatic and parotid salivary glands.5 Preganglionic neurons for the parasympathetic innervation of the sublingual and mandibular glands are provided by the parasympathetic nuclei of the facial nerve in the medulla.5 The preganglionic axons course in the facial nerve, through the tympanic cavity to join the lingual nerve, a branch of the mandibular nerve, at the level of the oval foramen. The oval foramen, located just medial to the temporomandibular joint, is also the foramen through which the mandibular nerve exits the cranial cavity. After the preganglionic axons join the mandibular nerve, they course to the mandibular and sublingual ganglia and synapse with parasympathetic ganglionic neurons. Parasympathetic postganglionic axon coursing with branches of the mandibular nerve ultimately innervate the mandibular and sublingual glands.5
Parasympathetic stimulation of the salivary glands mediated through acetylcholine is responsible for salivary production and flow. The sympathetic nervous system can modulate salivary secretion and composition through norepinephrine by stimulating the blood vessels and the acinar cells of the salivary glands.6 In general, sympathetic stimulation to the salivary glands results in vasoconstriction of the blood vessels, which reduces aqueous saliva and exocytosis of the acinar glandular cells. Overall, this results in a more proteinaceous and less voluminous saliva flow.6
An analysis of Max's salivation
In patients like Max with dysfunction of the trigeminal or the mandibular nerve, abnormal saliva that appears foamy, ropy and stringy accumulates in the oropharynx on the side ipsilateral to the nerve deficit (Figure 2).
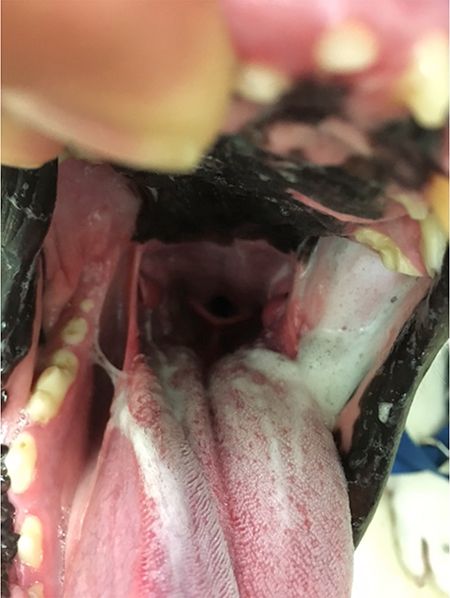
Figure 2. View of Max's oral cavity reveals the accumulation of thick, foamy saliva in the left caudal oropharynx.
The saliva buildup is located just caudal to the maxillary fourth premolar, which is where the openings to the oral cavity are for the parotid gland and zygomatic gland.7
How can one explain the accumulation of abnormal saliva? Given the normal function of cranial nerve VII (ability to blink the eye) and cranial nerve IX (ability to swallow normally), the preganglionic parasympathetic axons to the salivary glands are likely intact. Therefore, the abnormal composition and flow of saliva produced by the parotid and zygomatic salivary glands, as evidenced by accumulation of the thickened saliva in the caudal oropharynx, is most likely due to dysfunction of the postganglionic axons that course with the branches of mandibular nerve.
In cases like Max's, we hypothesize that the dysfunction of the trigeminal or mandibular nerve also affects the postganglionic parasympathetic axons that course alongside the axons of the trigeminal or mandibular nerve to reach the salivary glands. In other words, the loss of the conduit for the postganglionic axon provided by the trigeminal or mandibular nerve results in dysfunction of the parasympathetic stimulation to the salivary glands, which alters salivary flow and composition. Normally, the parasympathetic nervous system is responsible for the production of voluminous amounts of aqueous saliva. Parasympathetic denervation results in proteinaceous saliva that is more viscous than normal. This is appreciated clinically as an accumulation of thickened, ropy saliva that accumulates in the oropharynx on the side ipsilateral to trigeminal or mandibular nerve dysfunction.
The essentiality of salivation, and why it might go wrong
Normal salivation provides an important role in day-to-day life. Not only does it aid in food mastication and digestion but also in lubrication of the oral cavity, maintenance of normal pH of the mouth, and prevention of dental caries.8 Lack of salivation can cause significant oral discomfort and ulcerations as well as a detrimental decline in oral health.9 It's possible that with unilateral trigeminal nerve dysfunction and accumulation of abnormal saliva, there may be an increase in dental tartar on the side of the nerve dysfunction.7
A common cause of unilateral trigeminal nerve dysfunction is a nerve sheath neoplasm. Other causes of trigeminal nerve dysfunction include trauma, infectious or noninfectious neuritis, and other forms of neoplasia such as lymphoma. A complete physical and neurological examination may help narrow the differential diagnoses. Magnetic resonance imaging (MRI) of the head is the best way to establish a definitive diagnosis. With trigeminal nerve sheath neoplasms, MRI findings include an enlargement of the trigeminal nerve or its main branches, which display contrast enhancement, enlargement of the oval foramen from pressure necrosis from the expansile growth of the mandibular nerve, compression or invasion of the pons by the neoplasm, denervation atrophy of the masticatory muscles, and effusion in the tympanic cavity (middle ear) (Figure 3).
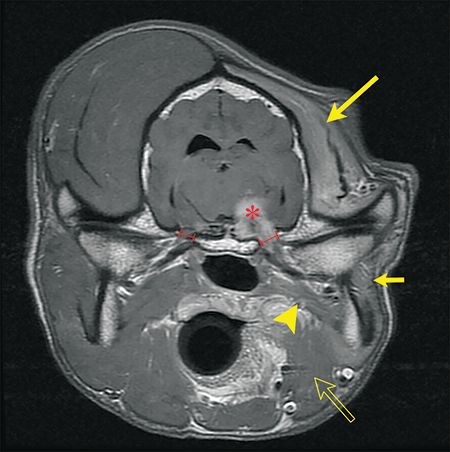
Figure 3. A transverse T1-weighted post-contrast MRI of Max's brain. A trigeminal nerve sheath neoplasm is compressing the brain stem (red asterisk) and is continuing along the mandibular branch as it exits via the oval foramen. Note the enlargement of the left oval foramen in comparison to the right. There is pronounced atrophy of the temporalis (large yellow arrow), masseter (small yellow arrow), digastricus (open yellow arrow) and pterygoid (yellow arrowhead).
What to do next
Treatment options for nerve sheath neoplasms include palliative therapy with anti-inflammatory drugs or definitive therapy with radiation therapy.10,11 Radiation therapy can provide long-term control.10
Given the potential for long-term survival after radiation therapy in dogs with trigeminal nerve sheath neoplasms, it's possible that the alteration in salivation may have a significant impact on the overall health and the quality of life of affected patients. It may be prudent to perform more frequent and thorough oral examinations to evaluate for pharyngeal function and salivary function in affected dogs. As the lack of autonomic functions to the head and face can lead to discomfort and secondary complications in our human patient counterparts, veterinarians should be aware of the potential for similar issues in dogs and cats.
Current accepted practices in dental care may provide a starting point for monitoring and planning dental procedures in affected patients.12 In the future, more tailored evaluation protocols, preventive care measures and therapeutic interventions may be developed to minimize the negative impact abnormal autonomic function has on oral and overall health and to maintain an excellent quality of life in affected patients.
References
1. Evans HE, de Lahunta A. Cranial nerves. In: Evans HE, de Lahunta A, eds. Miller's anatomy of the dog. Philadelphia: Elsevier Saunders, 2012;708-730.
2. Maggs DJ, Miller PE, Ofri R. Diseases of the lacrimal system. In: Maggs DJ, Miller PE, Ofri R, eds. Slatter's fundamentals of veterinary ophthalmology. 6th ed. St. Louis, Missouri: Elsevier, 2018;186-211.
3. Nederfors T. Xerostomia and hyposalivation. Adv Dent Res 2000;14:48-56.
4. Evans HE, de Lahunta A. The digestive apparatus and abdomen. In: Evans HE, de Lahunta A, eds. Miller's anatomy of the dog. Philadelphia: Elsevier Saunders, 2012;281-334.
5. Evans HE, de Lahunta A. The autonomic nervous system. In: Evans HE, de Lahunta A, eds. Miller's anatomy of the dog: Philadelphia: Elsevier Saunders, 2012;575-588.
6. Proctor GB, Carpenter GH. Regulation of salivary gland function by autonomic nerves. Auton Neurosci 2007;133:3-18.
7. Kent M, Glass EN, Song RB, et al. A salivation abnormality with trigeminal nerve dysfunction in dogs. J Vet Dent (in press).
8. Epstein JB, Scully C. The role of saliva in oral health and the causes and effects of xerostomia. J Can Dent Assoc 1992;58:217-221.
9. Einarson S, Gerdin EW, Hugoson A. Oral health-related quality of life and its relationship to self-reported oral discomfort and clinical status. Swed Dent J 2014;38:169-178.
10. Swift KE, McGrath S, Nolan MW, et al. Clinical and imaging findings, treatments, and outcomes in 27 dogs with imaging diagnosed trigeminal nerve sheath tumors: A multi-center study. Vet Radiol Ultrasound 2017;58:679-689.
11. Bagley R, Wheeler S, Klopp L, et al. Clinical features of trigeminal nerve-sheath tumor in 10 dogs. J Am Anim Hosp Assoc 1998;34:19-25.
12. Holmstrom SE, Bellows J, Juriga S, et al. 2013 AAHA dental care guidelines for dogs and cats. J Am Anim Hosp Assoc 2013;49:75-82.